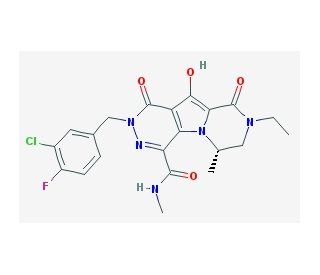
MK-2048 (CAS 869901-69-9)
QUICK LINKS
MK-2048 is a second generation integrase inhibitor for HIV-1 integrase. MK-2048 inhibits subtype B and subtype C integrase activities. MK-2048 inhibits R263K mutants slightly more effectively than G118R mutants. MK2048 displays substantially lower dissociation rates compared with raltegravir, another integrase inhibitor. MK-2048 is active against viruses resistant to RAL and EVG. MK-2048 exposure leads to the selection of G118R as a possible novel resistance mutation after 19 weeks. MK-2048, with continued pressure, subsequently leads to an additional substitution, at position E138K, after 29 weeks, within the IN gene. Although the G118R mutation alone confers only slight resistance to MK-2048 but not to RAL or EVG, its presence arouses a dramatic reduction in viral replication capacity compared to wild-type NL4-3. E138K both partially restores viral replication capacity and also contributes to increased levels of resistance against MK-2048.
MK-2048 (CAS 869901-69-9) References
- Anti-infectives: clinical progress of HIV-1 integrase inhibitors. | Al-Mawsawi, LQ., et al. 2008. Expert Opin Emerg Drugs. 13: 213-25. PMID: 18537517
- Comparative biochemical analysis of HIV-1 subtype B and C integrase enzymes. | Bar-Magen, T., et al. 2009. Retrovirology. 6: 103. PMID: 19906306
- Primary mutations selected in vitro with raltegravir confer large fold changes in susceptibility to first-generation integrase inhibitors, but minor fold changes to inhibitors with second-generation resistance profiles. | Goethals, O., et al. 2010. Virology. 402: 338-46. PMID: 20421122
- Identification of novel mutations responsible for resistance to MK-2048, a second-generation HIV-1 integrase inhibitor. | Bar-Magen, T., et al. 2010. J Virol. 84: 9210-6. PMID: 20610719
- Physical trapping of HIV-1 synaptic complex by different structural classes of integrase strand transfer inhibitors. | Pandey, KK., et al. 2010. Biochemistry. 49: 8376-87. PMID: 20799722
- Cross-resistance profile determination of two second-generation HIV-1 integrase inhibitors using a panel of recombinant viruses derived from raltegravir-treated clinical isolates. | Van Wesenbeeck, L., et al. 2011. Antimicrob Agents Chemother. 55: 321-5. PMID: 20956600
- HIV-1 integrase strand transfer inhibitors stabilize an integrase-single blunt-ended DNA complex. | Bera, S., et al. 2011. J Mol Biol. 410: 831-46. PMID: 21295584
- Integrase inhibitors effective against human T-cell leukemia virus type 1. | Seegulam, ME. and Ratner, L. 2011. Antimicrob Agents Chemother. 55: 2011-7. PMID: 21343468
- Resistance to raltegravir highlights integrase mutations at codon 148 in conferring cross-resistance to a second-generation HIV-1 integrase inhibitor. | Goethals, O., et al. 2011. Antiviral Res. 91: 167-76. PMID: 21669228
- Comparative antiviral activity of integrase inhibitors in human monocyte-derived macrophages and lymphocytes. | Scopelliti, F., et al. 2011. Antiviral Res. 92: 255-61. PMID: 21867733
- The HIV-1 integrase G118R mutation confers raltegravir resistance to the CRF02_AG HIV-1 subtype. | Malet, I., et al. 2011. J Antimicrob Chemother. 66: 2827-30. PMID: 21933786
- Dolutegravir maintains a durable effect against HIV replication in tissue culture even after drug washout. | Osman, N., et al. 2015. J Antimicrob Chemother. 70: 2810-5. PMID: 26142476
- Integrase Inhibitors: After 10 Years of Experience, Is the Best Yet to Come? | Brooks, KM., et al. 2019. Pharmacotherapy. 39: 576-598. PMID: 30860610
- Activation of PERK-ATF4-CHOP pathway as a novel therapeutic approach for efficient elimination of HTLV-1-infected cells. | Ikebe, E., et al. 2020. Blood Adv. 4: 1845-1858. PMID: 32369565
- Development and Evaluation of Nanoparticles-in-Film Technology to Achieve Extended In Vivo Exposure of MK-2048 for HIV Prevention. | Tong, X., et al. 2022. Polymers (Basel). 14: PMID: 35335526
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
MK-2048, 5 mg | sc-364535 | 5 mg | $405.00 | |||
MK-2048, 10 mg | sc-364535A | 10 mg | $555.00 |
