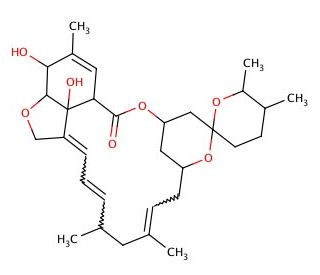
Molecular structure of Milbemycin A3, CAS Number: 51596-10-2
Milbemycin A3 (CAS 51596-10-2)
See product citations (1)
Application:
Milbemycin A3 is an antibiotic with acaricidal activity
CAS Number:
51596-10-2
Purity:
>95%
Molecular Weight:
528.70
Molecular Formula:
C31H44O7
Supplemental Information:
This is classified as a Dangerous Good for transport and may be subject to additional shipping charges.
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Product Citations
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Milbemycin A3 is an antibiotic derived from Streptomyces with acaricidal activity. The milbemycins are part of a family of macrocyclic lactones that are characterized by the spiroketal ring system that are produced by Streptomyces hygroscopicus subsp. aureolacrimosus and are suggested to stimulate chloride-gated ion channels. Milbemycin A3 and A4 (sc-202227) have been used to synthesize milbemycins alpha9, alpha10, alpha11, alpha12, alpha14, alpha15, alpha20, alpha21, alpha22, alpha23, alpha26, alpha7, delta(2,3), delta(4,26)-milbemycins A3, and A4.
Milbemycin A3 (CAS 51596-10-2) References
- Bioconversion of milbemycin-related compounds: isolation and utilization of non-producer, strain RNBC-5-51. | Nonaka, K., et al. 1999. J Antibiot (Tokyo). 52: 620-7. PMID: 10513841
- Synthesis of milbemycins beta9 and beta10 from milbemycins A3 and A4 and their biological activities. | Tsukiyama, T., et al. 2002. Biosci Biotechnol Biochem. 66: 1407-11. PMID: 12162570
- Synthesis of milbemycins alpha9, alpha10, alpha11, alpha12, alpha14, alpha15, alpha20, alpha21, alpha22, alpha23, alpha26, alpha27, delta(2,3),delta(4,26)-milbemycins A3, A4 from milbemycins A3, A4, and their acaricidal activities. | Tsukiyama, T., et al. 2002. J Antibiot (Tokyo). 55: 993-1003. PMID: 12546420
- Genetic engineering of Streptomyces bingchenggensis to produce milbemycins A3/A4 as main components and eliminate the biosynthesis of nanchangmycin. | Zhang, J., et al. 2013. Appl Microbiol Biotechnol. 97: 10091-101. PMID: 24077727
- Macrolide compounds, ivermectin and milbemycin D, stimulate chloride channels sensitive to GABAergic drugs in cultured chick spinal neurons. | Yamazaki, J., et al. 1989. Comp Biochem Physiol C Comp Pharmacol Toxicol. 93: 97-104. PMID: 2471607
- Two new α-class milbemycin metabolites from mutant Streptomyces avermitilis NEAU1069-3. | Li, LJ., et al. 2015. J Antibiot (Tokyo). 68: 354-6. PMID: 25424970
- Two new milbemycin derivatives from a genetically engineered strain Streptomyces bingchenggensis. | Li, JS., et al. 2021. J Asian Nat Prod Res. 23: 660-665. PMID: 32608247
- Modular polyketide synthase-derived insecticidal agents: from biosynthesis and metabolic engineering to combinatorial biosynthesis for their production. | Yi, JS., et al. 2023. Nat Prod Rep.. PMID: 36691749
- Sustainable chromatographic purification of milbemectin: Application of high-speed countercurrent chromatography coupled with off-line atmospheric pressure solid analysis probe-high resolution mass spectrometry. | Terajima, Y., et al. 2023. J Chromatogr A. 1694: 463901. PMID: 36889118
- Milbemycins, a new family of macrolide antibiotics: producing organism and its mutants. | Okazaki, T., et al. 1983. J Antibiot (Tokyo). 36: 438-41. PMID: 6853372
- Milbemycins, a new family of macrolide antibiotics. Fermentation, isolation and physico-chemical properties of milbemycins D, E, F, G, and H. | Takiguchi, Y., et al. 1983. J Antibiot (Tokyo). 36: 502-8. PMID: 6874568
- Milbemycins, a new family of macrolide antibiotics. Fermentation, isolation, physico-chemical properties and bioconversion of milbemycins J and K. | Ono, M., et al. 1983. J Antibiot (Tokyo). 36: 509-15. PMID: 6874569
- Milbemycins, a new family of macrolide antibiotics: fermentation, isolation and physico-chemical properties. | Takiguchi, Y., et al. 1980. J Antibiot (Tokyo). 33: 1120-7. PMID: 7451362
- Synthesis and anthelmintic activity of 13-alkoxymilbemycin derivatives. | Saito, A., et al. 1993. J Antibiot (Tokyo). 46: 1252-64. PMID: 8407588
- Microbial conversion of milbemycins: 28-hydroxylation of milbemycins by Amycolata autotrophica. | Nakagawa, K., et al. 1993. J Antibiot (Tokyo). 46: 518-9. PMID: 8478271
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Milbemycin A3, 1 mg | sc-202226 | 1 mg | $342.00 | |||
Milbemycin A3, 5 mg | sc-202226A | 5 mg | $1326.00 | |||
Milbemycin A3, 50 mg | sc-202226B | 50 mg | $6589.00 | |||
Milbemycin A3, 100 mg | sc-202226C | 100 mg | $12839.00 |
