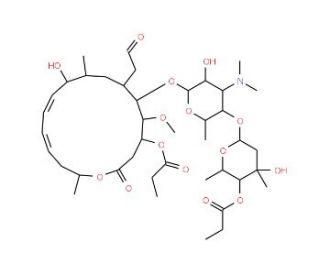
Molecular structure of Midecamycin, CAS Number: 35457-80-8
Midecamycin (CAS 35457-80-8)
CAS Number:
35457-80-8
Molecular Weight:
814
Molecular Formula:
C41H67NO15
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Midecamycin is an innovative anthracycline compound. Its mechanism of action involves the inhibition of DNA and RNA synthesis, leading to the demise of cancer cells. Additionally, Midecamycin induces apoptosis, or programmed cell death, specifically in cancer cells, intensifying its cytotoxic impact. Extensive research has investigated Midecamycin′s efficacy across various cell culture systems, revealing its preferential affinity for tumor cells over healthy cells.
Midecamycin (CAS 35457-80-8) References
- In vitro activity of midecamycin diacetate, a 16-membered macrolide, against Streptococcus pyogenes isolated in France, 1995-1999. | Schlegel, L., et al. 2001. Clin Microbiol Infect. 7: 362-6. PMID: 11531981
- [Acylation specificity of midecamycin 3-O-acyltransferase within Streptomyces spiramyceticus F21]. | Ma, C., et al. 2008. Sheng Wu Gong Cheng Xue Bao. 24: 2086-92. PMID: 19306580
- Direct electrochemistry and voltammetric determination of midecamycin at a multi-walled carbon nanotube coated gold electrode. | Wan, H., et al. 2011. Colloids Surf B Biointerfaces. 86: 247-50. PMID: 21543194
- [Diagnosis and treatment of acute tonsillopharyngitis. Consensus document update]. | Piñeiro Pérez, R., et al. 2020. An Pediatr (Engl Ed). 93: 206.e1-206.e8. PMID: 32605870
- Universal response method for the quantitative analysis of multi-components in josamycin and midecamycin using liquid chromatography coupled with charged aerosol detector. | Liu, G., et al. 2021. J Pharm Biomed Anal. 192: 113679. PMID: 33120309
- Midecamycin Is Inactivated by Several Different Sugar Moieties at Its Inactivation Site. | Lin, R., et al. 2021. Int J Mol Sci. 22: PMID: 34884439
- Controlling the Site Selectivity in Acylations of Amphiphilic Diols: Directing the Reaction toward the Apolar Domain in a Model Diol and the Midecamycin A1 Macrolide Antibiotic. | Fallek, R., et al. 2022. J Org Chem. 87: 9688-9698. PMID: 35801540
- Midecamycin in the treatment of otorhinolaryngological infections. | Polpathapee, S. and Na-Nakorn, A. 1985. J Med Assoc Thai. 68: 412-4. PMID: 4067459
- In vitro activity of midecamycin, a new macrolide antibiotic. | Neu, HC. 1983. Antimicrob Agents Chemother. 24: 443-4. PMID: 6639001
- Bacteriological evaluation of midecamycin acetate and its metabolites. | Yoshida, T., et al. 1982. Jpn J Antibiot. 35: 1462-74. PMID: 6982352
- Modifications of a macrolide antibiotic midecamycin. II. Reaction of midecamycin and 9-acetylmidecamycin with dimethylsulfoxide and acetic anhydride. | Inouye, S., et al. 1980. J Antibiot (Tokyo). 33: 61-71. PMID: 7372551
- Drug interaction between midecamycin and cyclosporin. | Finielz, P., et al. 1995. Nephron. 70: 136. PMID: 7617108
- Effects of ivermectin and midecamycin on ryanodine receptors and the Ca2+-ATPase in sarcoplasmic reticulum of rabbit and rat skeletal muscle. | Ahern, GP., et al. 1999. J Physiol. 514 (Pt 2): 313-26. PMID: 9852316
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Midecamycin, 25 mg | sc-295482 | 25 mg | $77.00 | |||
Midecamycin, 100 mg | sc-295482A | 100 mg | $153.00 |
