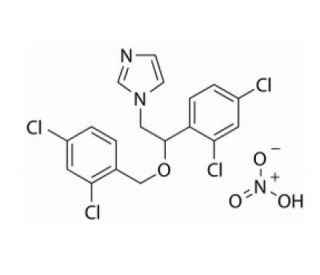
Molecular structure of Miconazole Nitrate, CAS Number: 22832-87-7
Miconazole Nitrate (CAS 22832-87-7)
See product citations (3)
Alternate Names:
NSC 169434; R 14889; (±)-Miconazole nitrate salt
Application:
Miconazole Nitrate is an antifungal agent that inhibits platelet cyclooxygenase
CAS Number:
22832-87-7
Purity:
≥98%
Molecular Weight:
479.15
Molecular Formula:
C18H14Cl4N2O•HNO3
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Product Citations
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Miconazole Nitrate is a topical antifungal agent found to inhibit platelet cyclooxygenase. Miconazole nitrate, a synthetic variation of the natural antifungal miconazole, serves as a topical solution for addressing fungal infections on the skin, nails, and mucous membranes. Available in cream, gel, and powder forms, this versatile agent effectively combats an array of skin conditions induced by fungi. These encompass athlete′s foot, jock itch, ringworm, and yeast infections. Furthermore, it proves beneficial in managing vaginal yeast infections and diaper rash.
Miconazole Nitrate (CAS 22832-87-7) References
- Miconazole nitrate in the treatment of dermatomycoses. | Degreef, H., et al. 1975. Dermatologica. 150: 103-10. PMID: 1158001
- Topical miconazole nitrate ointment in the treatment of diaper dermatitis complicated by candidiasis. | Spraker, MK., et al. 2006. Cutis. 77: 113-20. PMID: 16570675
- Development of miconazole nitrate containing chitosan microcapsules and their anti-Aspergillus niger activity. | Yuen, CW., et al. 2012. J Microencapsul. 29: 505-10. PMID: 22172026
- Miconazole nitrate bearing ultraflexible liposomes for the treatment of fungal infection. | Pandit, J., et al. 2014. J Liposome Res. 24: 163-9. PMID: 24479833
- Miconazole nitrate vaginal suppository 1,200 mg versus oral fluconazole 150 mg in treating severe vulvovaginal candidiasis. | Fan, S., et al. 2015. Gynecol Obstet Invest. 80: 113-8. PMID: 25720546
- Miconazole Nitrate-loaded Microparticles For Buccal Use: Immediate Drug Release and Antifungal Effect. | Cartagena, AF., et al. 2017. Curr Drug Deliv. 14: 1144-1153. PMID: 27719632
- Dermal miconazole nitrate nanocrystals - formulation development, increased antifungal efficacy & skin penetration. | Pyo, SM., et al. 2017. Int J Pharm. 531: 350-359. PMID: 28855137
- Development of Dermal Films Containing Miconazole Nitrate. | Bîrsan, M., et al. 2018. Molecules. 23: PMID: 29976876
- Influence of formulation variables on miconazole nitrate-loaded lipid based nanocarrier for topical delivery. | Al-Maghrabi, PM., et al. 2020. Colloids Surf B Biointerfaces. 193: 111046. PMID: 32416518
- Treatment methods for vulvovaginal candidiasis in pregnancy. | Bender, RA., et al. 2021. J Mycol Med. 31: 101138. PMID: 33971365
- Polymeric versus lipid nanocapsules for miconazole nitrate enhanced topical delivery: in vitro and ex vivo evaluation. | Abdel-Rashid, RS., et al. 2022. Drug Deliv. 29: 294-304. PMID: 35037528
- Physicochemical, rheological and antifungal evaluation of miconazole nitrate organogels for topical delivery. | Ambreen, Z., et al. 2022. Pak J Pharm Sci. 35: 1215-1221. PMID: 36218100
- Miconazole Nitrate Microparticles in Lidocaine Loaded Films as a Treatment for Oropharyngeal Candidiasis. | Tejada, G., et al. 2023. Materials (Basel). 16: PMID: 37176470
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Miconazole Nitrate, 1 g | sc-205753 | 1 g | $47.00 | |||
Miconazole Nitrate, 5 g | sc-205753A | 5 g | $158.00 |
