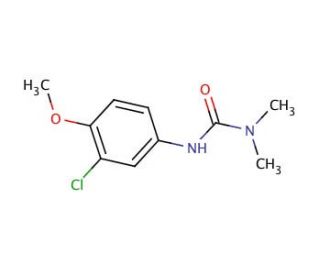
Molecular structure of Metoxuron, CAS Number: 19937-59-8
Metoxuron (CAS 19937-59-8)
CAS Number:
19937-59-8
Molecular Weight:
228.68
Molecular Formula:
C10H13ClN2O2
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Metoxuron is a herbicide that functions by inhibiting photosynthesis in plants. It acts by targeting the photosystem II complex in the chloroplasts of plant cells, disrupting the electron transport chain and ultimately leading to the production of reactive oxygen species. This disruption results in damage to the plant′s photosynthetic apparatus, leading to chlorosis and necrosis. Metoxuron′s mechanism of action involves binding to the QB-binding niche of the D1 protein in photosystem II, preventing the transfer of electrons and ultimately inhibiting the synthesis of ATP and NADPH. This disruption of the photosynthetic process ultimately leads to the death of the targeted plants.
Metoxuron (CAS 19937-59-8) References
- Multiresidue HPLC methods for phenyl urea herbicides in water. | Ruberu, SR., et al. 2000. J Agric Food Chem. 48: 4109-15. PMID: 10995323
- Phototransformation of metoxuron [3-(3-chloro-4-methoxyphenyl)-1,1-dimethylurea] in aqueous solution. | Boulkamh, A., et al. 2001. Pest Manag Sci. 57: 1119-26. PMID: 11802599
- Structure effect on the interaction of phenylurea herbicides with model biomembrane as an environmental mobility parameter. | Librando, V., et al. 2004. Environ Sci Technol. 38: 503-7. PMID: 14750726
- Clean up of phenylurea herbicides in plant sample extracts using molecularly imprinted polymers. | Tamayo, FG., et al. 2005. Anal Bioanal Chem. 381: 1234-40. PMID: 15756601
- Duckweed (Lemna gibba L.) as a test organism for homeopathic potencies. | Scherr, C., et al. 2007. J Altern Complement Med. 13: 931-7. PMID: 18047439
- Spectrophotometric assay of Isoproturon and Metoxuron in technical grade and formulation samples using 3-methylbenzothiozolin-2-one hydrazone hydrochloride. | Raju, KR., et al. 1992. Talanta. 39: 1387-90. PMID: 18965546
- Spectrophotometric determination of Isoproturon and Metoxuron using ethylacetoacetate and application to technical and formulation grade samples. | Ramakrishnam Raju, K., et al. 1996. Talanta. 43: 577-81. PMID: 18966521
- Assessment of the leaching potential of 12 substituted phenylurea herbicides in two agricultural soils under laboratory conditions. | Navarro, S., et al. 2012. J Agric Food Chem. 60: 5279-86. PMID: 22578198
- Leaching potential of phenylurea herbicides in a calcareous soil: comparison of column elution and batch studies. | Langeron, J., et al. 2014. Environ Sci Pollut Res Int. 21: 4906-13. PMID: 23097070
- Herbicide response polymorphisms in wild emmer wheat: ecological and isozyme correlations. | Nevo, E., et al. 1992. Theor Appl Genet. 84: 209-16. PMID: 24203049
- Photochemical transformation of phenylurea herbicides in surface waters: a model assessment of persistence, and implications for the possible generation of hazardous intermediates. | Fabbri, D., et al. 2015. Chemosphere. 119: 601-607. PMID: 25128892
- Urinary Pesticide Levels in Children and Adolescents Residing in Two Agricultural Communities in Mexico. | Sierra-Diaz, E., et al. 2019. Int J Environ Res Public Health. 16: PMID: 30781414
- Sampling and determination of fenitrothion, dimethoate, mevinphos, linuron, metoxuron and trifluralin from air. | Gudéhn, A. and Kolmodin-Hedman, B. 1987. J Chromatogr. 387: 420-7. PMID: 3558633
- Determination of phenylurea herbicide residues in vegetables by liquid chromatography after gel permeation chromatography and Florisil cartridge cleanup. | Sannino, A. 1998. J AOAC Int. 81: 1048-53. PMID: 9772748
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Metoxuron, 100 mg | sc-235879 | 100 mg | $61.00 |
