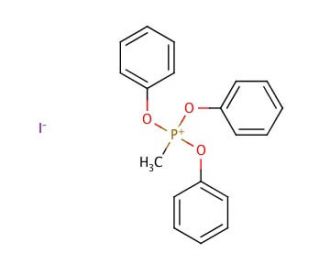
Methyltriphenoxyphosphonium Iodide (CAS 17579-99-6)
QUICK LINKS
Methyltriphenoxyphosphonium iodide (MTPPI) is a versatile organic iodide compound. It is a colorless, crystalline solid. Its utility extends to the synthesis of diverse heterocyclic compounds like pyrroles, furans, and thiophenes. Additionally, Methyltriphenoxyphosphonium iodide has proven valuable in the synthesis of polymers, including polystyrene, polyvinyl chloride, polyacrylonitrile, and polyethylene. Moreover, Methyltriphenoxyphosphonium iodide has found use as a reagent in the synthesis of diverse organic compounds, including amino acids, peptides, and carbohydrates. Its role in organic synthesis involves acting as a nucleophile, catalyzing a range of reactions. It exhibits Lewis acid behavior, activating electrophiles, while also functioning as a Bronsted acid, activating nucleophiles. Additionally, it can act as a base, activating electrophiles, and as a proton donor, activating nucleophiles.
Methyltriphenoxyphosphonium Iodide (CAS 17579-99-6) References
- Synthesis and structure of cyclic phosphate, phosphoramidate, phosphonates, and phosphonium salts. Phosphatrane formation. | Chandrasekaran, A., et al. 2000. Inorg Chem. 39: 5683-9. PMID: 11151368
- Synthesis of 5'-deoxy-5'-thioguanosine-5'-monophosphorothioate and its incorporation into RNA 5'-termini. | Zhang, B., et al. 2001. Org Lett. 3: 275-8. PMID: 11430053
- Total synthesis of ent-cholesterol via a steroid C,D-ring side-chain synthon. | Jiang, X. and Covey, DF. 2002. J Org Chem. 67: 4893-900. PMID: 12098302
- Synthesis of 9-fluorenemethyl boranophosphonodiphosphate via an H-phosphonate approach. | Liu, H., et al. 2007. Nucleosides Nucleotides Nucleic Acids. 26: 1455-7. PMID: 18066805
- Design and synthesis of a G-protein-coupled receptor antagonist library of aryloxyalkanolamines using a polymer-supported acyclic acetal linker. | Roberge, JY., et al. 2009. J Comb Chem. 11: 72-82. PMID: 19086798
- Biocompatible artificial DNA linker that is read through by DNA polymerases and is functional in Escherichia coli. | El-Sagheer, AH., et al. 2011. Proc Natl Acad Sci U S A. 108: 11338-43. PMID: 21709264
- Design, Synthesis, and Characterization of Sulfamide and Sulfamate Nucleotidomimetic Inhibitors of hHint1. | Shah, R., et al. 2016. ACS Med Chem Lett. 7: 780-4. PMID: 27563403
- Locked nucleic acid (LNA) enhances binding affinity of triazole-linked DNA towards RNA. | Kumar, P., et al. 2017. Chem Commun (Camb). 53: 8910-8913. PMID: 28748236
- Synthesis of the Halichondrin C1-C15 Fragment from a Halaven C27-C35 Byproduct: Stereospecific Intramolecular Kishi Reduction. | Kim, DS., et al. 2018. Org Lett. 20: 4295-4297. PMID: 29956939
- Dehydration reactions in polyfunctional natural products. | Hjerrild, P., et al. 2020. Nat Prod Rep. 37: 1043-1064. PMID: 32319503
- Solid-Phase Synthesis of RNA 5'-Azides and Their Application for Labeling, Ligation, and Cyclization Via Click Chemistry. | Warminski, M., et al. 2020. Curr Protoc Nucleic Acid Chem. 82: e112. PMID: 32716612
- Late-Stage Functionalisation of Peptides on the Solid Phase by an Iodination-Substitution Approach. | Werner, M., et al. 2022. Chemistry. 28: e202201339. PMID: 35700354
- Halo sugar nucleosides. 3. Reactions for the chlorination and bromination of nucleoside hydroxyl groups. | Verheyden, JP. and Moffatt, JG. 1972. J Org Chem. 37: 2289-99. PMID: 5040377
- Fluorinated pyrimidine nucleosides. 4. Synthesis and antitumor testing of a series of 2',5'-dideoxy- and 2',3',5'-trideoxynucleosides of 5-fluorouracil. | Cook, AF., et al. 1980. J Med Chem. 23: 852-7. PMID: 6447210
- Stereoselective synthesis, chemistry and antiviral evaluation of 1-(2,3-dideoxy-3-C-hydroxymethyl-beta-D-threo-pentofuranosyl)thymine derivatives. | Wengel, J., et al. 1995. Bioorg Med Chem. 3: 1223-9. PMID: 8564413
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Methyltriphenoxyphosphonium Iodide, 5 g | sc-207884 | 5 g | $82.00 |
