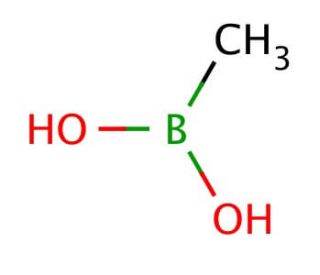
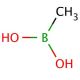
Methylboronic acid (CAS 13061-96-6)
QUICK LINKS
Methylboronic acid is a compound that functions as a Lewis acid in various chemical reactions. It acts as a catalyst in organic synthesis, particularly in the formation of carbon-carbon and carbon-oxygen bonds. The acid′s mode of action involves its ability to coordinate with electron-rich species, facilitating the activation of substrates and promoting the formation of new chemical bonds. In this capacity, it plays a role in the development of new methodologies and the production of complex organic molecules. Methylboronic acid′s reactivity with a range of functional groups makes it useful for the construction of diverse molecular architectures. Its ability to participate in cross-coupling reactions and other transformations contributes to the advancement of chemical research and the discovery of novel compounds.
Methylboronic acid (CAS 13061-96-6) References
- Gas chromatography/combustion/isotope-ratio-monitoring mass spectrometric analysis of methylboronic derivatives of monosaccharides: a new method for determining natural 13C abundances of carbohydrates. | van Dongen, BE., et al. 2001. Rapid Commun Mass Spectrom. 15: 496-500. PMID: 11268134
- Selective monomethylation of anilines by Cu(OAc)2-promoted cross-coupling with MeB(OH)2. | González, I., et al. 2009. Org Lett. 11: 1677-80. PMID: 19354317
- Preparation and synthetic application of partially protected brassinosteroids. | Khripach, VA., et al. 2010. Steroids. 75: 27-33. PMID: 19786042
- Determination of the stable carbon isotopic compositions of 2-methyltetrols in ambient aerosols from the Changbai Mountains. | Li, L., et al. 2010. Rapid Commun Mass Spectrom. 24: 1625-8. PMID: 20486258
- The spectroscopic (FTIR, FT-Raman, NMR and UV), first-order hyperpolarizability and HOMO-LUMO analysis of methylboronic acid. | Rani, U., et al. 2012. Spectrochim Acta A Mol Biomol Spectrosc. 92: 67-77. PMID: 22446752
- Aerobic Copper-Catalyzed O-Methylation with Methylboronic Acid. | Jacobson, CE., et al. 2015. J Org Chem. 80: 7305-10. PMID: 26111825
- New evidences on efficacy of boronic acid-based derivatization method to identify sugars in plant material by gas chromatography-mass spectrometry. | Faraco, M., et al. 2016. Talanta. 159: 40-46. PMID: 27474277
- Copper promoted N-alkylation of sulfoximines with alkylboronic acid under mild conditions. | Gupta, S., et al. 2017. Org Biomol Chem. 15: 8493-8498. PMID: 28952646
- Methylboronic acid fertilization alleviates boron deficiency symptoms in Arabidopsis thaliana. | Duran, C., et al. 2018. Planta. 248: 221-229. PMID: 29700610
- Regioselective single pot C3-glycosylation of strophanthidol using methylboronic acid as a transient protecting group. | Tay, JH., et al. 2019. J Antibiot (Tokyo). 72: 437-448. PMID: 30948784
- Virtues of Volatility: A Facile Transesterification Approach to Boronic Acids. | Hinkes, SPA. and Klein, CDP. 2019. Org Lett. 21: 3048-3052. PMID: 31012586
- Synthesis of Mono-N-Methyl Aromatic Amines from Nitroso Compounds and Methylboronic Acid. | Roscales, S. and Csákÿ, AG. 2019. ACS Omega. 4: 13943-13953. PMID: 31497712
- Facile deprotection of F-BODIPYs using methylboronic acid. | Smith, CD. and Thompson, A. 2020. RSC Adv. 10: 24273-24279. PMID: 35516207
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Methylboronic acid, 1 g | sc-253040 | 1 g | $41.00 |
