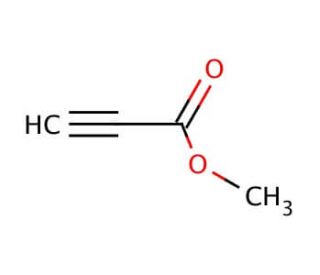
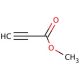
Methyl propiolate (CAS 922-67-8)
QUICK LINKS
Methyl propiolate is a chemical compound that functions as an alkylating agent in development applications. It acts by forming covalent bonds with nucleophilic groups in proteins and nucleic acids, leading to modifications in their structure and function. Methyl Propiolate′s mechanism of action involves the addition of alkyl groups to biomolecules, which can disrupt their normal biological activities. Methyl propiolate is known to react with thiol groups in proteins, leading to the formation of thioesters and disulfide bonds. Methyl Propiolate′s ability to alkylate biomolecules may be a useful for studying the effects of covalent modifications on biological systems. In experimental applications, methyl propiolate′s alkylating properties provide insights into the mechanisms of biomolecular interactions and the consequences of covalent modifications on cellular function.
Methyl propiolate (CAS 922-67-8) References
- Arylazide cycloaddition to methyl propiolate: DFT-based quantitative prediction of regioselectivity. | Molteni, G. and Ponti, A. 2003. Chemistry. 9: 2770-4. PMID: 12866540
- Synthesis of substituted chromenes through the DABCO-catalyzed reaction of but-3-yn-2-one and methyl propiolate with salicyl N-tosylimines (DABCO = 1,4-diazabicyclo[2.2.2]octane). | Shi, YL. and Shi, M. 2006. Chemistry. 12: 3374-8. PMID: 16470561
- Methyl 1-benzyl-1H-1,2,3-triazole-4-carboxyl-ate. | Huang, CC., et al. 2010. Acta Crystallogr Sect E Struct Rep Online. 66: o1690. PMID: 21587913
- Reactivity of 16-electron half-sandwich cobalt compounds containing a chelating 1,2-dicarba-closo-dodecaborane-1,2-dithiolate ligand towards methyl propiolate and dithio ligands. | Zhang, R., et al. 2012. Dalton Trans. 41: 12054-63. PMID: 22911065
- Facile synthesis of functionalized tetrahydroquinolines via domino Povarov reactions of arylamines, methyl propiolate and aromatic aldehydes. | Sun, J., et al. 2012. Beilstein J Org Chem. 8: 1839-43. PMID: 23209520
- Determination of glutathione in baker's yeast by capillary electrophoresis using methyl propiolate as derivatizing reagent. | Tsardaka, EC., et al. 2013. J Chromatogr A. 1300: 204-8. PMID: 23726088
- Methyl propiolate and 3-butynone: Starting points for synthesis of amphiphilic 1,2,3-triazole peptidomimetics for antimicrobial evaluation. | Bakka, TA., et al. 2017. Bioorg Med Chem. 25: 5380-5395. PMID: 28797773
- Influence of the Nucleophilic Ligand on the Reactivity of Carbonyl Rhenium(I) Complexes towards Methyl Propiolate: A Computational Chemistry Perspective. | Álvarez, D., et al. 2020. Molecules. 25: PMID: 32927650
- Introducing electrical conductivity to metal-organic framework thin films by templated polymerization of methyl propiolate. | Sen, B., et al. 2020. Nanoscale. 12: 24419-24428. PMID: 33300536
- Copper-Catalyzed β-Lactam Formation Initiated by 1,3-Azaprotio Transfer of Oximes and Methyl Propiolate. | Qi, Z. and Wang, S. 2021. Org Lett. 23: 5777-5781. PMID: 34240879
- Three-Component Reaction to 1,4,2-Diazaborole-Type Heteroarene Systems. | Li, J., et al. 2021. Angew Chem Int Ed Engl. 60: 27053-27061. PMID: 34597449
- Gold-Catalyzed Post-Polymerization Modification of Commodity Aromatic Polymers. | King, ER., et al. 2021. JACS Au. 1: 1342-1347. PMID: 34604843
- The synthesis of D-ribofuranosyl derivatives of methyl propiolate and a study of the activating influence of the ester group in cycloaddition reactions. | Buchanan, JG., et al. 1977. Carbohydr Res. 55: 225-38. PMID: 861978
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Methyl propiolate, 2.5 g | sc-255298 | 2.5 g | $39.00 | |||
Methyl propiolate, 25 g | sc-255298A | 25 g | $84.00 |
