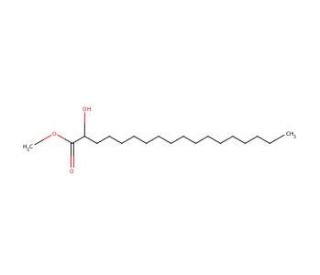
Methyl (±)-2-hydroxystearate (CAS 2420-35-1)
QUICK LINKS
Methyl (±)-2-hydroxystearate, a methyl ester derivative of (±)-2-hydroxystearic acid, has garnered significant attention in scientific research, particularly in the fields of lipid chemistry and material science. This compound is widely utilized as a model substrate in enzymology studies to investigate the catalytic activity and substrate specificity of lipases and esterases, which play crucial roles in lipid metabolism. Researchers leverage methyl (±)-2-hydroxystearate to explain the mechanistic details of enzyme-substrate interactions and the factors influencing enzyme kinetics, providing valuable insights into lipid digestion, absorption, and metabolism. Furthermore, methyl (±)-2-hydroxystearate serves as a precursor in the synthesis of biodegradable polymers and surfactants, where its long hydrocarbon chain and hydroxyl group confer desirable properties for various applications. Additionally, it finds utility in lipidomics, serving as an internal standard for quantifying fatty acid methyl esters in complex lipid mixtures, facilitating accurate lipid analysis. Its versatile roles in enzymology, synthetic chemistry, and lipid analysis highlight its significance as a fundamental tool in advancing our understanding of lipid-related processes and developing innovative applications in biochemical and materials research.
Methyl (±)-2-hydroxystearate (CAS 2420-35-1) References
- Cerebrosides and a monoacylmonogalactosylglycerol from Clinacanthus nutans. | Tuntiwachwuttikul, P., et al. 2004. Chem Pharm Bull (Tokyo). 52: 27-32. PMID: 14709863
- Pentafluorobenzoic anhydride as a derivatizing agent for alcohols and hydroxy fatty acid methyl esters detected by electron capture in gas chromatography. | Crabtree, DV., et al. 1989. J Chromatogr. 466: 251-70. PMID: 2501332
- Flavor Differences of Edible Parts of Grass Carp between Jingpo Lake and Commercial Market. | Chen, H., et al. 2022. Foods. 11: PMID: 36076779
- Fatty acid and long chain base composition of adrenal medulla gangliosides. | Ledeen, R. and Salsman, K. 1970. Lipids. 5: 751-6. PMID: 4318605
- Occurrence of phosphonosphingolipids in Bdellovibrio bacteriovorus strain UKi2. | Steiner, S., et al. 1973. J Bacteriol. 116: 1199-211. PMID: 4752938
- Gangliosides in subacute sclerosing leukoencephalitis: isolation and fatty acid composition of nine fractions. | Ledeen, R., et al. 1968. J Lipid Res. 9: 129-36. PMID: 5637421
- A study of the fatty acid metabolism of the yeast Pityrosporum ovale. | Wilde, PF. and Stewart, PS. 1968. Biochem J. 108: 225-31. PMID: 5691082
- Organic Sulfur Derivatives. V.2 Preparation and Properties of Some Long-Chain Mercapto Acids and Related Compounds3 | Koenig, N. H., GEORGE S. SASIN, and Daniel Swern. 1958. the Journal of Organic Chemistry. 23: 1525–1530.
- Chlorophyll-sensitized peroxidation of saturated fatty acid esters | Sastry, Y. Sita Rama, and Gollamudi Lakshminarayana. 1971. Journal of the American Oil Chemists' Society. 48: 452–454.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Methyl (±)-2-hydroxystearate, 10 mg | sc-215312 | 10 mg | $73.00 |
