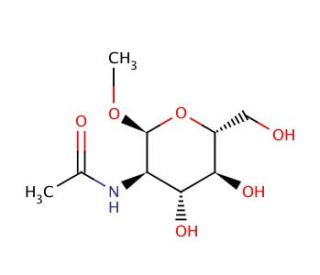
Methyl N-acetyl-α-D-glucosaminide (CAS 6082-04-8)
QUICK LINKS
Methyl N-acetyl-α-D-glucosaminide is a derivative of sugar employed as a fundamental component in organic synthesis. It has been instrumental in the creation of a broad range of substances that include agrochemicals, materials, and various types of compounds like carbohydrates, peptides, and proteins. Its unique properties and adaptability have led to its increased usage in organic synthesis in recent years. Beyond its role in synthesis, M2ADG is recognized for its contribution to scientific research. It′s known to serve as a substrate for enzymes like glycosyltransferases and glycosidases, making it useful in studies focused on the structure and functions of proteins, the development of new compounds, carbohydrate metabolism, and the synthesis of glycoproteins. The enzymes glycosyltransferases and glycosidases are known to catalyze the transfer of a sugar moiety from M2ADG to another molecule, such as a protein or lipid. This reaction is typically reversible, resulting in a glycosylated molecule.
Methyl N-acetyl-α-D-glucosaminide (CAS 6082-04-8) References
- New GlcNAc/GalNAc-specific lectin from the ascidian Didemnum ternatanum. | Molchanova, V., et al. 2005. Biochim Biophys Acta. 1723: 82-90. PMID: 15784180
- Methyl 2-acetamido-2-deoxy-β-D-glucopyranoside dihydrate and methyl 2-formamido-2-deoxy-β-D-glucopyranoside. | Hu, X., et al. 2011. Acta Crystallogr C. 67: o146-50. PMID: 21467623
- Scope and limitations of carbohydrate hydrolysis for de novo glycan sequencing using a hydrogen peroxide/metallopeptide-based glycosidase mimetic. | Peng, T., et al. 2018. Carbohydr Res. 458-459: 85-88. PMID: 29475194
- N-Acetyl-D-glucosamine-specific lectin from the ascidian Didemnum ternatanum. | Belogortseva, N., et al. 1998. Biochim Biophys Acta. 1380: 249-56. PMID: 9565695
- Studies on Amino-hexoses. VII The N-Deacetylation of Methyl N-Acetyl α-D-Glucosaminide | Fujinaga Mamoru, Matsushima Yoshio. 1964. Bulletin of the Chemical Society of Japan. 37, 4: 468-470.
- Periodate Oxidation of Some Carbohydrates as Examined by NMR Spectroscopy | and Shigehiro Hirano, Tamotsu Fukuda, Motonori Sato. 1974. Agricultural and Biological Chemistry. 38, 12: 2539–2543.
- Biotransformations in carbohydrates synthesis. N-Acetylgalactosaminyl transfer on to methyl N-acetyl-β-D-glucosaminide (methyl 2-acetamido-2-deoxy-β-D-glucopyranoside) and methyl N-acetyl-α-D-glucosaminide (methyl 2-acetamido-2-deoxy-α-D-glucopyranoside) catalysed by a β-N-acetylgalactosaminidase from Aspergillus oryzae | David H. G. Crout, Suddham Singh, Ben E. P. Swoboda, Peter Critchley and Walter T. Gibson. 1992. J. Chem. Soc., Chem. Commun. 9: 704-705.
- 3‐and 4‐Uloses Derived from N‐Acetyl‐d‐glucosamine: A Unique Pair of Complementary Organocatalysts for Asymmetric Epoxidation of Alkenes | Schoeberl, C., & Jaeger, V. 2012. Advanced Synthesis & Catalysis. 354(5): 790-796.
- Decadal nitrogen addition alters chemical composition of soil organic matter in a boreal forest | S Hasegawa, J Marshall, T Sparrman, T Näsholm. 2021. Geoderma. 386: 114906.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Methyl N-acetyl-α-D-glucosaminide, 100 mg | sc-221930 | 100 mg | $398.00 |
