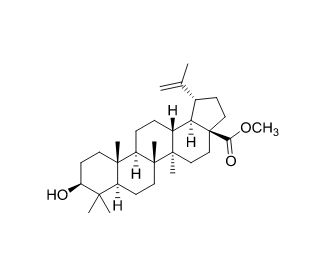
Molecular structure of Methyl betulinate, CAS Number: 2259-06-5
Methyl betulinate (CAS 2259-06-5)
Alternate Names:
Betulinic acid methyl ester; Mairin methyl ester; Methyl betulate
Application:
Methyl betulinate is a pentacyclic lupane terpene
CAS Number:
2259-06-5
Purity:
≥95%
Molecular Weight:
470.74
Molecular Formula:
C31H50O3
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Methyl betulinate, a lupane-type triterpenoid, is an esterified form of betulinic acid that has been discovered in Helicteres hirsuta and exhibits a wide range of biological activities. It demonstrates inhibitory effects on HIV-1 reverse transcriptase, with an IC50 value of 11 μM. Its cytotoxicity has been observed in various cell lines, including SK-LU-1, HepG2, HeLa, SK-MEL-2, and AGS, with IC50 values of 60.84 μg/ml, 77.43 μg/ml, 80.17 μg/ml, 66.17 μg/ml, and 69.94 μg/ml, respectively.
Methyl betulinate (CAS 2259-06-5) References
- Inhibitory effect of some triterpenes from cacti on 32Pi-incorporation into phospholipids of HeLa cells promoted by 12-O-tetradecanoylphorbol-13-acetate. | Kinoshita, K., et al. 1999. Phytomedicine. 6: 73-7. PMID: 10374243
- Identification of three triterpenoids in almond hulls. | Takeoka, G., et al. 2000. J Agric Food Chem. 48: 3437-9. PMID: 10956130
- Erythrocyte membrane modifying agents and the inhibition of Plasmodium falciparum growth: structure-activity relationships for betulinic acid analogues. | Ziegler, HL., et al. 2004. Bioorg Med Chem. 12: 119-27. PMID: 14697777
- Anti-inflammatory and antitumor-promoting effects of the triterpene acids from the leaves of Eriobotrya japonica. | Banno, N., et al. 2005. Biol Pharm Bull. 28: 1995-9. PMID: 16204964
- Pharmacological properties of the ubiquitous natural product betulin. | Alakurtti, S., et al. 2006. Eur J Pharm Sci. 29: 1-13. PMID: 16716572
- Glycosidation of lupane-type triterpenoids as potent in vitro cytotoxic agents. | Gauthier, C., et al. 2006. Bioorg Med Chem. 14: 6713-25. PMID: 16787747
- Lupane-type triterpenoids from Microtropis fokienensis and Perrottetia arisanensis and the apoptotic effect of 28-hydroxy-3-oxo-lup-20(29)-en-30-al. | Chen, IH., et al. 2008. J Nat Prod. 71: 1352-7. PMID: 18590313
- Anti-leishmanial activity of betulin derivatives. | Alakurtti, S., et al. 2010. J Antibiot (Tokyo). 63: 123-6. PMID: 20139867
- Bioactive constituents from Harpephyllum caffrum Bernh. and Rhus coriaria L. | Shabana, MM., et al. 2011. Pharmacogn Mag. 7: 298-306. PMID: 22262932
- Synthesis and antiplasmodial activity of betulinic acid and ursolic acid analogues. | Innocente, AM., et al. 2012. Molecules. 17: 12003-14. PMID: 23085651
- Chemistry of Hyptis mutabilis: new pentacyclic triterpenoids. | Pereda-Miranda, R. and Gascón-Figueroa, M. 1988. J Nat Prod. 51: 996-8. PMID: 3204385
- Jujube metabolome selection determined the edible properties acquired during domestication. | Zhang, Z., et al. 2022. Plant J. 109: 1116-1133. PMID: 34862996
- Studies on anti-inflammatory agents. V. A new anti-inflammatory constituent of Pyracantha crenulata roem. | Otsuka, H., et al. 1981. Chem Pharm Bull (Tokyo). 29: 3099-104. PMID: 7337925
Inhibitor of:
SCAP.Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Methyl betulinate, 1 mg | sc-506423 | 1 mg | $156.00 |
