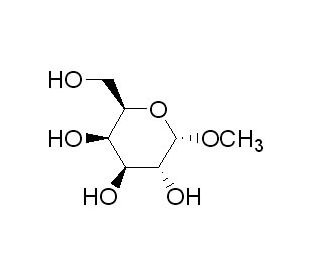
Methyl α-D-galactopyranoside (CAS 3396-99-4)
QUICK LINKS
Methyl α-D-galactopyranoside is a chemical compound used as a reference in carbohydrate chemistry and as a substrate in enzymatic studies. The compound serves as a model to understand the stereochemistry and reactivity of glycosidic bonds in sugars, due to its α-anomeric configuration. In enzymology, Methyl α-D-galactopyranoside is frequently employed to probe the specificity and activity of glycoside hydrolases, such as α-galactosidases, which are enzymes that catalyze the cleavage of α-galactosidic linkages. The compound is also useful in the study of glycosylation processes, where it can act as a donor molecule in the synthesis of oligosaccharides and glycoconjugates. Additionally, researchers utilize Methyl α-D-galactopyranoside in the development of chromatographic techniques for the separation and analysis of carbohydrates. Its role in the synthesis of glycomimetics, molecules that mimic the structure of sugars, is another aspect of research, contributing to the understanding of carbohydrate-protein interactions.
Methyl α-D-galactopyranoside (CAS 3396-99-4) References
- Neuraminidase substrate promiscuity permits a mutant Micromonospora viridifaciens enzyme to synthesize artificial carbohydrates. | Cheng, LL., et al. 2014. Biochemistry. 53: 3982-89. PMID: 24870444
- Synthesis and evaluation of iminocoumaryl and coumaryl derivatized glycosides as galectin antagonists. | Rajput, VK., et al. 2014. Bioorg Med Chem Lett. 24: 3516-20. PMID: 24973029
- Snake venom galactoside-binding lectins: a structural and functional overview. | Sartim, MA. and Sampaio, SV. 2015. J Venom Anim Toxins Incl Trop Dis. 21: 35. PMID: 26413085
- Synthesis of Conformationally-Locked cis- and trans-Bicyclo[4.4.0] Mono-, Di-, and Trioxadecane Modifications of Galacto- and Glucopyranose; Experimental Limiting 3JH,H Coupling Constants for the Estimation of Carbohydrate Side Chain Populations and Beyond. | Amarasekara, H., et al. 2018. J Org Chem. 83: 881-897. PMID: 29241001
- The binding of boronated peptides to low affinity mammalian saccharides. | Kowalczyk, W., et al. 2018. Biopolymers.. PMID: 29381187
- Scope and limitations of carbohydrate hydrolysis for de novo glycan sequencing using a hydrogen peroxide/metallopeptide-based glycosidase mimetic. | Peng, T., et al. 2018. Carbohydr Res. 458-459: 85-88. PMID: 29475194
- Repurposed Drugs That Block the Gonococcus-Complement Receptor 3 Interaction Can Prevent and Cure Gonococcal Infection of Primary Human Cervical Epithelial Cells. | Poole, J., et al. 2020. mBio. 11: PMID: 32127453
- Targeting Oligosaccharides and Glycoconjugates Using Superselective Binding Scaffolds. | Tommasone, S., et al. 2020. Adv Funct Mater. 30: 2002298. PMID: 32774200
- Synthesis of Tetravalent Thio- and Selenogalactoside-Presenting Galactoclusters and Their Interactions with Bacterial Lectin PA-IL from Pseudomonas aeruginosa. | Illyés, TZ., et al. 2021. Molecules. 26: PMID: 33494330
- Distinguishing Galactoside Isomers with Mass Spectrometry and Gas-Phase Infrared Spectroscopy. | Ho, JS., et al. 2021. J Am Chem Soc. 143: 10509-10513. PMID: 34236183
- Targeting the Central Pocket of the Pseudomonas aeruginosa Lectin LecA. | Siebs, E., et al. 2022. Chembiochem. 23: e202100563. PMID: 34788491
- Synthesis of an Azide- and Tetrazine-Functionalized [60]Fullerene and Its Controlled Decoration with Biomolecules. | Gulumkar, V., et al. 2022. ACS Omega. 7: 1329-1336. PMID: 35036794
- Novel Galactopyranoside Esters: Synthesis, Mechanism, In Vitro Antimicrobial Evaluation and Molecular Docking Studies. | Matin, P., et al. 2022. Molecules. 27: PMID: 35807371
- Biochemical and ligand binding properties of recombinant Xenopus laevis cortical granule lectin-1. | Deetanya, P., et al. 2022. Heliyon. 8: e10396. PMID: 36061023
- Natural Compounds of Lasia spinosa (L.) Stem Potentiate Antidiabetic Actions by Regulating Diabetes and Diabetes-Related Biochemical and Cellular Indexes. | Rashid, MM., et al. 2022. Pharmaceuticals (Basel). 15: PMID: 36558918
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Methyl α-D-galactopyranoside, 5 g | sc-221923B | 5 g | $80.00 | |||
Methyl α-D-galactopyranoside, 25 g | sc-221923 | 25 g | $287.00 | |||
Methyl α-D-galactopyranoside, 100 g | sc-221923A | 100 g | $865.00 |
