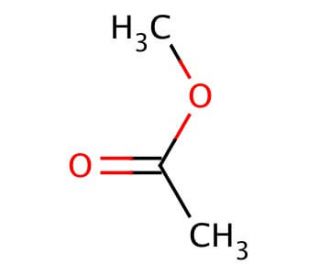
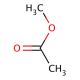
Methyl acetate (CAS 79-20-9)
QUICK LINKS
Methyl acetate is a versatile organic compound that has been extensively studied in various research contexts. Its chemical structure, comprising a methyl group attached to an acetyl moiety, allows it to participate in a range of chemical reactions. In the context of organic synthesis, methyl acetate serves as a precursor to the production of other compounds, such as esters and lactones, through nucleophilic substitution and condensation reactions. Additionally, methyl acetate has been utilized as a solvent and reactant in the synthesis of polymers, including polyurethanes and polyesters. In the field of materials science, methyl acetate has been investigated as a precursor to the production of novel nanomaterials, such as metal-organic frameworks (MOFs), which exhibit unique properties due to their porous structures. Furthermore, methyl acetate has been employed as a reactant in the synthesis of bioactive compounds, such as chalcones and flavonoids, which have been shown to exhibit antimicrobial and antioxidant properties.
Methyl acetate (CAS 79-20-9) References
- Phytogenic biosynthesis and emission of methyl acetate. | Jardine, K., et al. 2014. Plant Cell Environ. 37: 414-24. PMID: 23862653
- Methyl Acetate Synthesis by Esterification on the Modified Ferrierite: Correlation of Acid Sites Measured by Pyridine IR and NH3-TPD for Steady-State Activity. | Park, JH., et al. 2016. J Nanosci Nanotechnol. 16: 4626-30. PMID: 27483801
- Thermal Decomposition of Potential Ester Biofuels. Part I: Methyl Acetate and Methyl Butanoate. | Porterfield, JP., et al. 2017. J Phys Chem A. 121: 4658-4677. PMID: 28517940
- Continuous production of biodiesel under supercritical methyl acetate conditions: Experimental investigation and kinetic model. | Farobie, O. and Matsumura, Y. 2017. Bioresour Technol. 241: 720-725. PMID: 28622654
- Are Methanol-Derived Foliar Methyl Acetate Emissions a Tracer of Acetate-Mediated Drought Survival in Plants? | Dewhirst, RA., et al. 2021. Plants (Basel). 10: PMID: 33672332
- Methyl acetate, a highly volatile floral semiochemical mediating specialized plant-beetle interactions. | Maia, ACD., et al. 2021. Naturwissenschaften. 108: 21. PMID: 33934251
- Stepwise Iodide-Free Methanol Carbonylation via Methyl Acetate Activation by Pincer Iridium Complexes. | Yoo, C. and Miller, AJM. 2021. J Am Chem Soc. 143: 12633-12643. PMID: 34347476
- Theoretical study about the hydrogen abstraction reactions on methyl acetate on combustion conditions. | da Silva Pereira, L. and Baptista, L. 2022. J Mol Model. 28: 226. PMID: 35869168
- Infrared intensities of methyl acetate, an interstellar compound - comparisons of three organic esters. | Yarnall, YY. and Hudson, RL. 2022. Spectrochim Acta A Mol Biomol Spectrosc. 283: 121738. PMID: 35985234
- Methyl Acetate Boosts the Low-Temperature Performance of Li4Ti5O12/Graphite Dual-Ion Batteries. | Mei, W., et al. 2024. Langmuir. 40: 11541-11547. PMID: 38767530
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Methyl acetate, 1 ml | sc-211875 | 1 ml | $34.00 | |||
Methyl acetate, 5 ml | sc-211875A | 5 ml | $93.00 |
