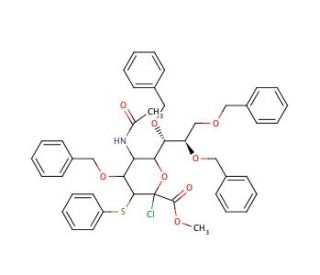
Methyl 5-acetamido-4,7,8,9-tetra-O-benzyl-2-chloro-2,3,5-trideoxy-3-phenylthio-D-erytho-β-L-gluco-2-nonulopyranosonate
QUICK LINKS
Methyl 5-acetamido-4,7,8,9-tetra-O-benzyl-2-chloro-2,3,5-trideoxy-3-phenylthio-D-erythro-β-L-gluco-2-nonulopyranosonate is a compound extensively utilized in glycosylation reactions for the synthesis of complex carbohydrates and glycoconjugates. Its mechanism of action involves its role as a glycosyl donor in carbohydrate chemistry. The chloro group facilitates glycosylation reactions by participating in the formation of glycosidic bonds with appropriate glycosyl acceptors. With the benzyl groups serving as protecting groups for hydroxyl functionalities, this compound enables precise control over regioselectivity during glycosylation reactions. Its utility in carbohydrate chemistry research extends to the synthesis of oligosaccharides with diverse structures, which are invaluable for studying carbohydrate-protein interactions, cell surface glycans, and carbohydrate-based materials. Moreover, Methyl 5-acetamido-4,7,8,9-tetra-O-benzyl-2-chloro-2,3,5-trideoxy-3-phenylthio-D-erythro-β-L-gluco-2-nonulopyranosonate is employed in the construction of glycan microarrays, which are powerful tools for high-throughput screening of carbohydrate-protein interactions. Its precise structure and functionality make it a cornerstone in carbohydrate chemistry research, driving advancements in glycobiology, chemical biology, and materials science.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Methyl 5-acetamido-4,7,8,9-tetra-O-benzyl-2-chloro-2,3,5-trideoxy-3-phenylthio-D-erytho-β-L-gluco-2-nonulopyranosonate, 5 mg | sc-224084 | 5 mg | $360.00 |
