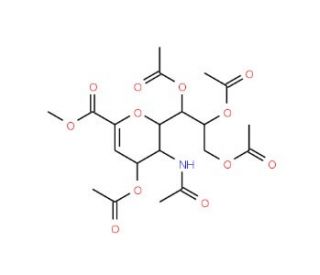
Methyl 5,7,8,9-Tetra-O-acetyl-4-acylamino-2,6-anhydro-3,4-dideoxy-D-glycero-D-galacto-2-enonate
QUICK LINKS
Methyl 5,7,8,9-Tetra-O-acetyl-4-acylamino-2,6-anhydro-3,4-dideoxy-D-glycero-D-galacto-2-enonate is a complex synthetic carbohydrate derivative utilized primarily in advanced organic synthesis and carbohydrate chemistry research. This compound′s structure is characterized by multiple acetyl groups that protect the hydroxyl functionalities, a 4-acylamino group which introduces nitrogen functionality into the sugar ring, and an anhydro formation that affects the ring′s electronic and steric properties. Its unique dideoxy and enonate features make it particularly valuable for studying the chemical behavior under various reaction conditions, including nucleophilic attacks and rearrangements. The chemical is extensively used in the study of glycosylation reactions where it acts as a glycosyl donor, allowing researchers to explore new pathways for bond formation and understand the mechanisms by which sugars interact with other biomolecules. It is instrumental in developing methodologies for the synthesis of glycopeptides and glyconjugates, crucial for exploring cellular communication processes and molecular recognition. The inclusion of the acylamino group enhances the compound′s utility in probing the effects of nitrogen-containing substituents on the reactivity and stability of glycosidic linkages, providing insights into the synthesis of novel carbohydrate-based materials with potential applications in fields like materials science and biotechnology.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Methyl 5,7,8,9-Tetra-O-acetyl-4-acylamino-2,6-anhydro-3,4-dideoxy-D-glycero-D-galacto-2-enonate, 1 mg | sc-224085 | 1 mg | $430.00 |
