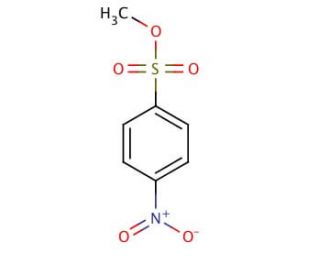
Methyl 4-nitrobenzenesulfonate (CAS 6214-20-6)
QUICK LINKS
Methyl 4-nitrobenzenesulfonate functions as an electrophilic aromatic substitution reagent in organic synthesis. It acts as a sulfonating agent, introducing a sulfonate group onto aromatic compounds. The mechanism of action involves the nitro group of the molecule serving as a strong electron-withdrawing group, making the aromatic ring more susceptible to electrophilic attack. This allows for the substitution of the nitro group with the aromatic ring, resulting in the formation of the sulfonate group. The reaction typically occurs under acidic conditions, facilitating the activation of the aromatic ring for sulfonation. Methyl 4-Nitrobenzenesulfonate′s function in organic synthesis involves the modification of aromatic compounds, enabling the introduction of sulfonate groups for further functionalization.
Methyl 4-nitrobenzenesulfonate (CAS 6214-20-6) References
- Influence of the addition of alcohol on the reaction methyl-4-nitrobenzenesulfonate + Br(-) in tetradecyltrimethylammonium bromide aqueous micellar solutions. | Muñoz, M., et al. 2003. J Colloid Interface Sci. 266: 208-14. PMID: 12957601
- Study of the reaction between methyl 4-nitrobenzenesulfonate and bromide ions in mixed single-chain-gemini micellar solutions: kinetic evidence for morphological transitions. | del Mar Graciani, M., et al. 2008. J Colloid Interface Sci. 328: 324-30. PMID: 18840382
- Modular ipso/ ortho Difunctionalization of Aryl Bromides via Palladium/Norbornene Cooperative Catalysis. | Dong, Z., et al. 2018. J Am Chem Soc. 140: 8551-8562. PMID: 29906109
- Investigation of drug release modulation from poly(2-oxazoline) micelles through ultrasound. | Salgarella, AR., et al. 2018. Sci Rep. 8: 9893. PMID: 29967422
- Molecular Mechanisms Responsible for Pharmacological Effects of Genipin on Mitochondrial Proteins. | Kreiter, J., et al. 2019. Biophys J. 117: 1845-1857. PMID: 31706565
- Processing of (Co)Poly(2-oxazoline)s by Electrospinning and Extrusion from Melt and the Postprocessing Properties of the (Co)Polymers. | Wałach, W., et al. 2020. Polymers (Basel). 12: PMID: 32024273
- Cold atmospheric pressure plasma: simple and efficient strategy for preparation of poly(2-oxazoline)-based coatings designed for biomedical applications. | Šrámková, P., et al. 2020. Sci Rep. 10: 9478. PMID: 32528062
- Poly(2-oxazoline) Matrices with Temperature-Dependent Solubility-Interactions with Water and Use for Cell Culture. | Oleszko-Torbus, N., et al. 2020. Materials (Basel). 13: PMID: 32545841
- Selective Partial Hydrolysis of 2-isopropyl-2-oxazoline Copolymers towards Decreasing the Ability to Crystallize. | Oleszko-Torbus, N., et al. 2020. Materials (Basel). 13: PMID: 32752250
- Thermal Behaviour of Common Thermoresponsive Polymers in Phosphate Buffer and in Its Salt Solutions. | Otulakowski, Ł., et al. 2020. Polymers (Basel). 13: PMID: 33379398
- Alternative to Poly(2-isopropyl-2-oxazoline) with a Reduced Ability to Crystallize and Physiological LCST. | Wałach, W., et al. 2021. Int J Mol Sci. 22: PMID: 33672348
- Effect of Dexamethasone on Thermoresponsive Behavior of Poly(2-Oxazoline) Diblock Copolymers. | Majerčíková, M., et al. 2021. Polymers (Basel). 13: PMID: 33919321
- Quantitative Determination of Four Potential Genotoxic Impurities in the Active Pharmaceutical Ingredients in TSD-1 Using UPLC-MS/MS. | Wang, T., et al. 2022. Molecules. 27: PMID: 35807373
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Methyl 4-nitrobenzenesulfonate, 1 g | sc-250361 | 1 g | $73.00 |
