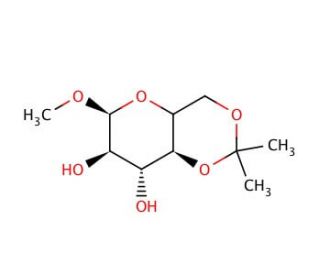
Methyl 4,6-O-Isopropylidene-α-D-mannopyranoside (CAS 63167-67-9)
QUICK LINKS
Methyl 4,6-O-Isopropylidene-α-D-mannopyranoside is a chemically modified derivative of mannose, extensively utilized in carbohydrate chemistry for its protective isopropylidene group that stabilizes the sugar molecule during synthetic transformations. This compound features a methyl group at the anomeric position, making it an important building block in glycosylation reactions, where it is used to construct more complex carbohydrate structures. In scientific research, this molecule is particularly valuable for its role in studying the stereochemical outcomes of glycosylation reactions. The isopropylidene group at the 4,6 positions protects adjacent hydroxyl groups from unwanted chemical reactions, allowing chemists to manipulate other parts of the sugar molecule with greater specificity and control. This specificity is crucial for synthesizing precise glycosidic linkages required in the creation of diverse oligosaccharides, which are used to probe biological processes such as cell signaling and molecular recognition. Moreover, Methyl 4,6-O-Isopropylidene-α-D-mannopyranoside is utilized in the development of chiral intermediates in synthetic organic chemistry. Its configuration and protective groups facilitate the creation of asymmetric centers, which are essential for the production of biologically active molecules. The research involving this compound significantly contributes to advancing the fields of glycomics and synthetic chemistry, offering insights into the complex interactions of carbohydrates and their role in biological systems.
Methyl 4,6-O-Isopropylidene-α-D-mannopyranoside (CAS 63167-67-9) References
- New evidence for the mechanism of the tin(II) chloride catalyzed reactions of vicinal diols with diazodiphenylmethane in 1,2-dimethoxyethane. | Pétursson, S. 2001. Carbohydr Res. 331: 239-45. PMID: 11383893
- Facile syntheses of the hexasaccharide repeating unit of the exopolysaccharide from Cryptococcus neoformans serovar A. | Zhang, J. and Kong, F. 2003. Bioorg Med Chem. 11: 4027-37. PMID: 12927865
- Efficient synthesis of a 3,6-branched mannose hepta- and octasaccharide. | Zhang, J., et al. 2003. Carbohydr Res. 338: 2039-46. PMID: 14505870
- Sequential directed epoxydation-acidolysis from glycals with MCPBA. A flexible approach to protected glycosyl donors. | Marín, I., et al. 2011. J Org Chem. 76: 9622-9. PMID: 21913684
- Tuneable regioselectivity during the mono-etherification of the 2,3-diol of a mannose derivative. | Petursson, S., et al. 2014. Carbohydr Res. 388: 37-43. PMID: 24607539
- Regioselective monoalkylations of the vicinal cis-diol group in mannopyranosides using diaryldiazoalkanes-tin(II) chloride | Petursson, S., & Webber, J. M. 1982. Carbohydrate Research. 103(1): 41-52.
- Protecting groups in carbohydrate chemistry | Pétursson, S. 1997. Journal of chemical education. 74(11): 1297.
- Parasite glycoconjugates. Part 11. 1 Preparation of phosphodisaccharide synthetic probes, substrate analogues for the elongating α-D-mannopyranosylphosphate transferase in the Leishmania | Ross, A. J., Ivanova, I. A., Ferguson, M. A., & Nikolaev, A. V. 2001. Journal of the Chemical Society, Perkin Transactions 1. (1): 72-81.
- An efficient synthesis of a hexasaccharide-the repeating unit of the exopolysaccharide from Cryptococcus neoformans serovar A | Zhang, J., & Kong, F. 2003. Tetrahedron letters. 44(9): 1839-1842.
- Diarylmethyl ethers for the protection of polyols | Petursson, S. 2013. Journal of Chemistry. 2013(1): 183049.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Methyl 4,6-O-Isopropylidene-α-D-mannopyranoside, 1 g | sc-221920 | 1 g | $300.00 |
