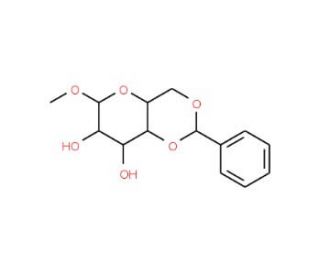
Methyl 4,6-O-Benzylidene-β-D-glucopyranoside (CAS 14155-23-8)
QUICK LINKS
Methyl 4,6-O-Benzylidene-β-D-glucopyranoside is a carbohydrate derivative known for its distinct benzylidene acetal protection at the 4 and 6 positions, which is crucial for synthetic applications and structural analysis in glycoscience. This compound is commonly used as a glycosyl donor or acceptor in carbohydrate synthesis due to the protective benzylidene group, which provides stability and facilitates regioselective deprotection. The benzylidene acetal allows selective cleavage to expose hydroxyl groups at desired positions, enabling researchers to synthesize glycosides or oligosaccharides with specific glycosidic linkages. By serving as a versatile scaffold, this compound helps develop stereochemically defined carbohydrate structures, which are vital in understanding enzyme specificity, protein-carbohydrate interactions, and complex carbohydrate biosynthesis. Methyl 4,6-O-Benzylidene-β-D-glucopyranoside also provides insights into glycosylation mechanisms, as it mimics structural motifs present in natural glycans. It is frequently utilized in studying glycosylation reactions′ stereoselectivity, particularly in the formation of β-glycosidic bonds. Furthermore, its stable benzylidene acetal has made it valuable in enzyme assays and mechanistic studies involving glycosidases and glycosyltransferases, contributing to the development of glycosylation methodologies. Thus, this compound continues to be instrumental in carbohydrate chemistry research by providing a controlled platform for studying glycosylation and synthesizing complex oligosaccharides.
Methyl 4,6-O-Benzylidene-β-D-glucopyranoside (CAS 14155-23-8) References
- Further evidence for the gelation ability-structure correlation in sugar-based gelators. | Gronwald, O., et al. 2001. Carbohydr Res. 331: 307-18. PMID: 11383900
- Organometallic supramolecular chemistry with monosaccharides: triethylammonium mu-chloro-bis[chloro(eta5-cyclopentadienyl)-(methyl 4,6-o-benzylidene-beta-D-glucopyranosidato-1kappaO2,1:2kappaO3) zirconate]. | Jessen, L., et al. 2001. Chemistry. 7: 3791-7. PMID: 11575781
- Synthesis of oxidized methyl 4-O-methyl-beta-D-glucopyranoside and methyl beta-D-glucopyranosyl-(1-->4)-beta-D-glucopyranoside derivatives as substrates for fluorescence labeling reactions. | Röhrling, J., et al. 2002. Carbohydr Res. 337: 691-700. PMID: 11950465
- Hydroxymethyl rotamer populations in disaccharides. | Roën, A., et al. 2003. J Org Chem. 68: 4615-30. PMID: 12790564
- Silver(I) oxide mediated selective monoprotection of diols in pyranosides. | Wang, H., et al. 2004. J Org Chem. 69: 5774-7. PMID: 15307757
- Synthesis of monodeoxy and mono-O-methyl congeners of methyl beta-D-mannopyranosyl-(1-->2)-beta-D-mannopyranoside for epitope mapping of anti-Candida albicans antibodies. | Nycholat, CM. and Bundle, DR. 2009. Carbohydr Res. 344: 1397-411. PMID: 19679227
- H-bonding activation in highly regioselective acetylation of diols. | Zhou, Y., et al. 2013. J Org Chem. 78: 11618-22. PMID: 24164588
- Semiconducting Fabrics by In Situ Topochemical Synthesis of Polydiacetylene: A New Dimension to the Use of Organogels. | Krishnan, BP., et al. 2016. Angew Chem Int Ed Engl. 55: 2345-9. PMID: 26840212
- Site-Selective Acylation of Pyranosides with Oligopeptide Catalysts. | Seitz, A., et al. 2021. J Org Chem. 86: 3907-3922. PMID: 33617252
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Methyl 4,6-O-Benzylidene-β-D-glucopyranoside, 2 g | sc-358347 | 2 g | $245.00 |
