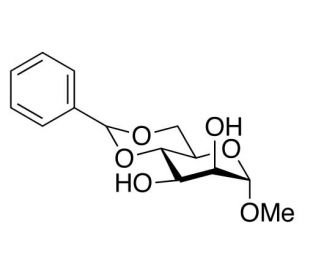
Methyl 4,6-O-Benzylidene-α-D-mannopyranoside (CAS 4148-58-7)
QUICK LINKS
Methyl 4,6-O-Benzylidene-α-D-mannopyranoside is a synthetic carbohydrate derivative used primarily in the field of organic chemistry and enzymology for studying carbohydrate-active enzymes. This compound features a benzylidene acetal protecting group at the 4 and 6 positions, which stabilizes the glycosidic bond against acidic hydrolysis, making it a suitable substrate for studies on selective enzymatic reactions involving glycosidases and glycosyltransferases. In research, Methyl 4,6-O-Benzylidene-α-D-mannopyranoside is utilized to explore the specificity and mechanism of action of mannosidases, enzymes that catalyze the hydrolysis of mannose-containing glycosides. This understanding is crucial for developing synthetic pathways for the assembly of mannose-containing oligosaccharides, which have significant applications in studying cell surface interactions and glycoprotein functions in various biological systems. Additionally, this compound is used in kinetic studies to quantify enzyme activity and to investigate the factors affecting the catalysis in model systems. The research involving this chemical extends to its role in the synthesis of complex carbohydrates, where its protected form allows for the precise formation of glycosidic linkages without premature breakdown. These capabilities are essential for advancing glycoscience, particularly in understanding how glycosidic bonds influence biological recognition and signaling processes.
Methyl 4,6-O-Benzylidene-α-D-mannopyranoside (CAS 4148-58-7) References
- Synthesis of alpha-Manp-(1-->2)-alpha-Manp-(1-->3)-alpha-Manp-(1-->3)-Manp, the tetrasaccharide repeating unit of Escherichia coli O9a, and alpha-Manp-(1-->2)-alpha-Manp-(1-->2)-alpha-Manp-(1-->3)-alpha-Manp-(1-->3)-Manp, the pentasaccharide repeating unit of E. coli O9 and Klebsiella O3. | Chen, L., et al. 2002. Carbohydr Res. 337: 383-90. PMID: 11861011
- Complexes of sodium vanadate(V) with methyl alpha-D-mannopyranoside, methyl alpha- and beta-D-galactopyranoside, and selected O-methyl derivatives: a 51V and 13C NMR study. | Noleto, GR., et al. 2003. Carbohydr Res. 338: 1745-50. PMID: 12892941
- Synthesis of a hexasaccharide fragment of the O-deacetylated GXM of C. neoformans serotype B. | Zhao, W. and Kong, F. 2004. Carbohydr Res. 339: 1779-86. PMID: 15220088
- Facile synthesis of the heptasaccharide repeating unit of O-deacetylated GXM of C. neoformans serotype B. | Zhao, W. and Kong, F. 2005. Bioorg Med Chem. 13: 121-30. PMID: 15582457
- Simple, unambiguous syntheses of 2,3,6- and 2,4,6-tri-O-methyl-D-mannose. | Choy, YM. and Unrau, AM. 1971. Carbohydr Res. 17: 439-43. PMID: 5150906
- Synthesis and reactions of unsaturated sugars: IV. Methyl 4, 6-O-benzylidene-α-d-erythro-hex-2-enopyranoside and its hydrolysis by acid. | Albano, et al. 1966. Carbohydrate Research. 2.5: 349-362.
- Quantitative treatment of lanthanide-induced shifts for some carbohydrate systems where chelation is observed. | McArdle, Patrick, et al. 1981. Carbohydrate Research. 90.2: 165-172.
- Regioselective monoalkylations of the vicinal cis-diol group in mannopyranosides using diaryldiazoalkanes-tin (II) chloride. | Petursson, et al. 1982. Carbohydrate Research. 103.1: 41-52.
- Hydrogenolysis of some methyl 4, 6-O-benzylidene-α-d-mannopyranoside derivatives: unambiguous synthesis of the 2, 4-and 3, 4-dimethyl ethers of methyl α-d-mannopyranoside. | Noumi, Kazuhiko, et al. 1984. Carbohydrate research. 134.1: 172-176.
- New mannose-derived ketones as organocatalysts for enantioselective dioxirane-mediated epoxidation of arylalkenes. Part 3: Chiral ketones from sugars. | Vega-Pérez and José M., et al. 2011. Tetrahedron. 67.37: 7057-7065.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Methyl 4,6-O-Benzylidene-α-D-mannopyranoside, 1 g | sc-211865 | 1 g | $340.00 |
