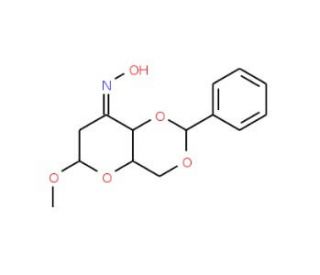
Methyl 4,6-O-Benzylidene-2-deoxy-alpha-D-erythro-hexopyranosid-3-ulose Oxime (CAS 63598-32-3)
QUICK LINKS
Methyl 4,6-O-Benzylidene-2-deoxy-alpha-D-erythro-hexopyranosid-3-ulose oxime is a carbohydrate derivative featuring a 4,6-O-benzylidene protective group, a methylated anomeric carbon, and an oxime functionality at the 3-position. This unique combination of functional groups and the benzylidene protecting strategy offers a versatile framework for modifying glycosidic structures and synthesizing complex oligosaccharides. The oxime functionality specifically facilitates the investigation of carbohydrate oxime glycosylation reactions. In research, this compound is predominantly employed in carbohydrate chemistry to explore glycosylation mechanisms, particularly those involving oxime glycosides, which can form stable linkages with other carbohydrates or proteins. The 4,6-O-benzylidene group ensures selective reactivity by protecting adjacent hydroxyl groups while leaving the 2-position available for functionalization. The oxime group acts as a reactive site for nucleophilic substitutions or condensations, allowing the construction of glycosyl derivatives with nitrogen-based linkages. Scientists have also utilized this compound to study the stereochemistry of glycosidic bond formation and develop regioselective methods for synthesizing glycosylated structures. Its unique configuration serves as a model compound for elucidating carbohydrate-protein interactions, shedding light on glycosyltransferase enzyme specificity. The precise and controlled modifications enabled by this derivative are crucial in understanding carbohydrate recognition and developing carbohydrate-based probes for studying the role of glycans in cellular biology.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Methyl 4,6-O-Benzylidene-2-deoxy-alpha-D-erythro-hexopyranosid-3-ulose Oxime, 1 g | sc-295434 | 1 g | $550.00 |
