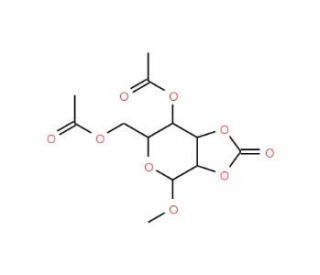
Methyl 4,6-Di-O-acetyl-β-D-mannopyranoside 2,3-carbonate
QUICK LINKS
Methyl 4,6-Di-O-acetyl-β-D-mannopyranoside 2,3-carbonate is an acylated carbohydrate derivative known for its unique structural modifications, which provide valuable insights into glycosylation processes and carbohydrate chemistry. This chemical is frequently employed in glycosyl donor studies due to its protected hydroxyl groups and the carbonate moiety that make it a suitable candidate for exploring selective glycosylation reactions. Researchers utilize it to investigate stereoselectivity in glycosidic bond formation, which is pivotal for understanding the synthesis of complex oligosaccharides. By employing this compound in various chemical reactions, scientists have studied the influence of protecting groups on glycosylation patterns, providing insights into designing selective carbohydrate modifications. Moreover, it aids in developing efficient synthetic methodologies that mimic naturally occurring oligosaccharides or glycoconjugates, crucial in exploring the structure-function relationships of carbohydrates. Additionally, this compound has served as a precursor in the synthesis of diverse sugar derivatives, facilitating research into enzyme specificity and activity by acting as a substrate in carbohydrate-active enzyme assays. The exploration of its reactivity offers significant contributions to the understanding of stereochemical outcomes in glycosylation and continues to be instrumental in carbohydrate synthesis research.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Methyl 4,6-Di-O-acetyl-β-D-mannopyranoside 2,3-carbonate, 500 mg | sc-358346 | 500 mg | $245.00 |
