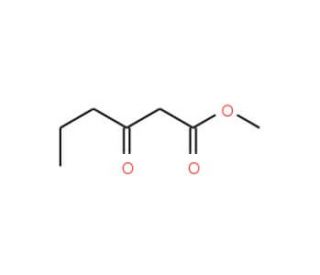
Methyl 3-Oxohexanoate (CAS 30414-54-1)
QUICK LINKS
Methyl 3-Oxohexanoate, with the CAS number 30414-54-1, is a methyl ester derivative of 3-oxohexanoic acid, featuring a ketone functional group on the third carbon of a six-carbon chain. This molecular structure, particularly the ketone group, plays a crucial role in its chemical behavior and applications in research. In synthetic organic chemistry, Methyl 3-Oxohexanoate is extensively utilized as a reagent for the synthesis of various complex molecules. The ketone group makes it highly reactive, suitable for condensation reactions that are fundamental in building cyclic compounds and for the synthesis of larger, more complex organic frameworks. The ester group in Methyl 3-Oxohexanoate also affords it versatility, being readily involved in transesterification reactions which are valuable for modifying ester functionalities into more useful forms, or for introducing new functional groups. In the realm of flavor and fragrance chemistry, this compound is investigated for its potential to contribute to or modify aromatic and flavor profiles, given its ability to undergo breakdown and recombination in flavor synthesis processes. Additionally, it serves as a useful model in studies of biodegradation of esters, helping to understand the environmental fate of similar compounds. Thus, Methyl 3-Oxohexanoate is a significant compound in chemical research, offering insights into synthetic strategies and environmental science.
Methyl 3-Oxohexanoate (CAS 30414-54-1) References
- Contemporary Strategies for the Synthesis of Tetrahydropyran Derivatives: Application to Total Synthesis of Neopeltolide, a Marine Macrolide Natural Product. | Fuwa, H. 2016. Mar Drugs. 14: PMID: 27023567
- Inhibition of Mycobacterium tuberculosis dihydrodipicolinate synthase by alpha-ketopimelic acid and its other structural analogues. | Shrivastava, P., et al. 2016. Sci Rep. 6: 30827. PMID: 27501775
- Inversion of cpADH5 Enantiopreference and Altered Chain Length Specificity for Methyl 3-Hydroxyalkanoates. | Ensari, Y., et al. 2017. Chemistry. 23: 12636-12645. PMID: 28727189
- Metabolites of the Anaerobic Degradation of n-Hexane by Denitrifying Betaproteobacterium Strain HxN1. | Küppers, J., et al. 2020. Chembiochem. 21: 373-380. PMID: 31294892
- Engineered P450 BM3 and cpADH5 coupled cascade reaction for β-oxo fatty acid methyl ester production in whole cells. | Ensari, Y., et al. 2020. Enzyme Microb Technol. 138: 109555. PMID: 32527525
- Suspect screening and targeted analysis of acyl coenzyme A thioesters in bacterial cultures using a high-resolution tribrid mass spectrometer. | Cakić, N., et al. 2021. Anal Bioanal Chem. 413: 3599-3610. PMID: 33881564
- Avenoleic acid: a new oxylipin from oat seeds. | Hamberg, M. 1997. Adv Exp Med Biol. 433: 69-72. PMID: 9561106
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Methyl 3-Oxohexanoate, 25 mg | sc-295422 | 25 mg | $43.00 |
