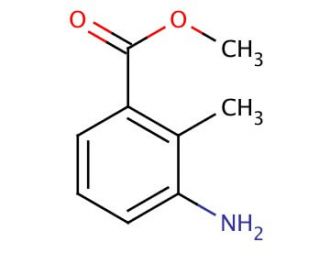
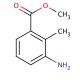
Methyl 3-amino-2-methylbenzoate (CAS 18583-89-6)
QUICK LINKS
Methyl 3-amino-2-methylbenzoate is an aromatic ester that is of interest in organic chemistry research, particularly in areas involving the synthesis of heterocyclic compounds. Due to the presence of both amino and ester functional groups, it is frequently used as a precursor or building block in the construction of more complex molecules. This compound is often involved in nucleophilic substitution reactions where the amino group can be a key site for reaction with various electrophiles. Additionally, in studies of reaction mechanisms, Methyl 3-amino-2-methylbenzoate serves to provide insights into the electronic effects of substituents on aromatic rings. Its use in the preparation of dyes, pigments, and other functionalized aromatics is also notable, aiding in the development of materials with specific optical properties.
Methyl 3-amino-2-methylbenzoate (CAS 18583-89-6) References
- Discovery and SAR of 5-(3-chlorophenylamino)benzo[c][2,6]naphthyridine-8-carboxylic acid (CX-4945), the first clinical stage inhibitor of protein kinase CK2 for the treatment of cancer. | Pierre, F., et al. 2011. J Med Chem. 54: 635-54. PMID: 21174434
- Atropisomeric 4-phenyl-4H-1,2,4-triazoles as selective glycine transporter 1 inhibitors. | Sugane, T., et al. 2013. J Med Chem. 56: 5744-56. PMID: 23837744
- From hit to lead: Structure-based discovery of naphthalene-1-sulfonamide derivatives as potent and selective inhibitors of fatty acid binding protein 4. | Gao, DD., et al. 2018. Eur J Med Chem. 154: 44-59. PMID: 29775936
- Design and Synthesis of Novel Amino-triazine Analogues as Selective Bruton's Tyrosine Kinase Inhibitors for Treatment of Rheumatoid Arthritis. | Kawahata, W., et al. 2018. J Med Chem. 61: 8917-8933. PMID: 30216722
- Discovery of a Potent and Orally Bioavailable Melatonin Receptor Agonist. | Hoashi, Y., et al. 2021. J Med Chem. 64: 3059-3074. PMID: 33682410
- Exploration of imatinib and nilotinib-derived templates as the P2-Ligand for HIV-1 protease inhibitors: Design, synthesis, protein X-ray structural studies, and biological evaluation. | Ghosh, AK., et al. 2023. Eur J Med Chem. 255: 115385. PMID: 37150084
- Recent advance of small-molecule drugs for clinical treatment of multiple myeloma. | Zhao, JH., et al. 2023. Eur J Med Chem. 257: 115492. PMID: 37210838
- Synthesis and biological properties of substituted 1,4-dihydro-5-methyl-4-oxo-3-quinolinecarboxylic acids. | Miyamoto, H., et al. 1995. Bioorg Med Chem. 3: 1699-706. PMID: 8770395
- Practical and efficient synthesis of the (R)-atropisomer of a 4-phenyl 1,2,4-triazole derivative as a selective GlyT1 inhibitor | Takashi Sugane, Noritaka Hamada, Takahiko Tobe, Wataru Hamaguchi, Itsuro Shimada, Kyoichi Maeno, Junji Miyata, Takeshi Suzuki, Tetsuya Kimizuka, Shuichi Sakamoto, Shin-ichi Tsukamoto. 2012. Tetrahedron: Asymmetry. 1528-1533: 23.
- Four new amide derivatives of pyridinecarboxylic acids. Synthesis, structure and spectroscopic characterization | Dorota Kwiatek, Maciej Kubicki, Piotr Barczyński, Stefan Lis, Zbigniew Hnatejko. 2017. Journal of Molecular Structure. 1145: 86-93.
- A constructive synthetic approach for reduction of nitro heterocyclics- characterization and degradation studies | Anindita Chatterjee a, Garbham Venkata Sivaprasad a, Ravi Kumar Devarakonda b, Ramachandrula Krishna Kumar b, Bavireddi Basaveswara Rao b, Gajjala Krishna Reddy a, Ramayanam Satya Kameswara Sharma a. 2023. Journal of the Indian Chemical Society. 100.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Methyl 3-amino-2-methylbenzoate, 25 g | sc-228522 | 25 g | $193.00 |
