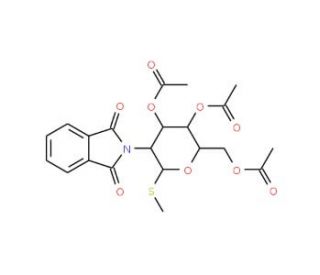
Methyl 3,4,6-Tri-O-acetyl-2-deoxy-2-phthalimido-1-thio-beta-D-glucopyranoside (CAS 79528-48-6)
QUICK LINKS
Methyl 3,4,6-Tri-O-acetyl-2-deoxy-2-phthalimido-1-thio-beta-D-glucopyranoside is a versatile carbohydrate derivative frequently employed in glycoscience research. It has a thio-glycosidic bond that enhances its role as a glycosyl donor, while the protective acetyl groups safeguard hydroxyls at positions 3, 4, and 6. The 2-deoxy-2-phthalimido substitution at position 2, a known amino group protector, provides stability and enables specific reactions. The thio group at the anomeric position increases resistance to hydrolysis, allowing this compound to be utilized in the synthesis of oligosaccharides. Researchers often use it to introduce nitrogen-based linkages due to the 2-phthalimido group, which can be converted to free amino groups after deprotection. This feature helps construct amino sugar-based glycoconjugates that mimic naturally occurring structures. The compound is instrumental in synthesizing N-linked oligosaccharides that are crucial for studying the glycosylation of proteins. Its beta-thio configuration is particularly valuable in ensuring stereoselective glycosylation, which is essential in investigating the role of glycan structure in molecular recognition. This derivative also provides a starting point for synthesizing glycosylated probes, aiding researchers in elucidating glycosyltransferase enzyme mechanisms and protein-carbohydrate interactions. Overall, it is a fundamental building block in carbohydrate chemistry due to its reliable functionality and adaptability.
Methyl 3,4,6-Tri-O-acetyl-2-deoxy-2-phthalimido-1-thio-beta-D-glucopyranoside (CAS 79528-48-6) References
- Synthesis of a di-, tri-, and tetra-saccharide unit of the group B streptococcal common antigen. | Pozsgay, V. and Jennings, HJ. 1988. Carbohydr Res. 179: 61-75. PMID: 3061650
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Methyl 3,4,6-Tri-O-acetyl-2-deoxy-2-phthalimido-1-thio-beta-D-glucopyranoside, 1 g | sc-295423 | 1 g | $294.00 | |||
Methyl 3,4,6-Tri-O-acetyl-2-deoxy-2-phthalimido-1-thio-beta-D-glucopyranoside, 5 g | sc-295423A | 5 g | $1105.00 |
