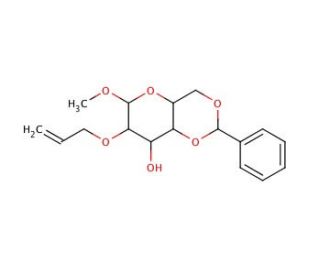

Methyl 2-O-Allyl-4,6-O-benzylidene-α-D-mannopyranoside (CAS 82228-09-9)
QUICK LINKS
Methyl 2-O-Allyl-4,6-O-benzylidene-α-D-mannopyranoside, a derivative of mannose, serves as a valuable tool in carbohydrate chemistry and glycobiology research due to its unique structural features and reactivity. This compound is commonly employed in the synthesis of complex carbohydrates and glycoconjugates, particularly in the context of glycan array fabrication, where precise control over glycan structure and functionality is essential for probing molecular interactions with lectins, antibodies, and other carbohydrate-binding proteins. Additionally, Methyl 2-O-Allyl-4,6-O-benzylidene-α-D-mannopyranoside has been utilized as a substrate or precursor in enzymatic glycosylation reactions, facilitating the enzymatic synthesis of oligosaccharides and glycosylated compounds with tailored structures. Its allyl and benzylidene functionalities provide strategic handles for selective chemical modification, enabling the introduction of diverse functional groups or tags for subsequent conjugation or immobilization onto surfaces in biosensing applications. Furthermore, this compound has been investigated for its potential role as a glycosyl donor or acceptor in enzymatic and chemical glycosylation strategies, offering insights into glycosylation mechanisms and substrate specificity of glycosyltransferases. Overall, Methyl 2-O-Allyl-4,6-O-benzylidene-α-D-mannopyranoside serves as a versatile building block in carbohydrate chemistry, facilitating the synthesis of structurally diverse glycans and glycoconjugates for various research applications in glycobiology, biochemistry, and biotechnology.
Methyl 2-O-Allyl-4,6-O-benzylidene-α-D-mannopyranoside (CAS 82228-09-9) References
- Synthesis of model oligosaccharides of biological significance. 3. Synthesis of carbon‐13 labelled trimannosides | Winnik, F. M., Carver, J. P., & Krepinsky, J. J. 1983. Journal of Labelled Compounds and Radiopharmaceuticals. 20(8): 983-989.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Methyl 2-O-Allyl-4,6-O-benzylidene-α-D-mannopyranoside, 250 mg | sc-221897 | 250 mg | $300.00 |
