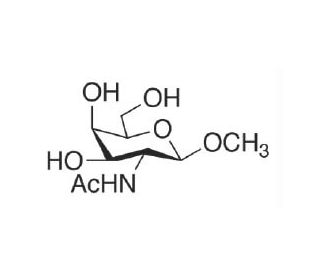
Methyl 2-Acetamido-2-deoxy-β-D-galactopyranoside (CAS 3055-46-7)
QUICK LINKS
Methyl 2-Acetamido-2-deoxy-β-D-galactopyranoside is a synthetic compound widely employed in carbohydrate chemistry research for its role in the synthesis of complex oligosaccharides and glycoconjugates. Its chemical structure, featuring an acetamido group and a deoxy sugar moiety, provides unique reactivity and selectivity in glycosylation reactions. Researchers utilize this compound to explore various synthetic strategies for constructing structurally diverse carbohydrate molecules with precise control over regiochemistry and stereochemistry. Furthermore, methyl 2-Acetamido-2-deoxy-β-D-galactopyranoside serves as a valuable tool in elucidating the mechanisms of glycosylation reactions, including the role of protecting groups in controlling glycosidic bond formation and the stereochemical outcomes of glycosylation reactions. Additionally, this compound finds applications in biochemical and biophysical studies, such as the development of glycoconjugates for investigating carbohydrate-protein interactions or the synthesis of carbohydrate-based materials for biomaterials research. Its versatility and utility in carbohydrate chemistry research contribute to advancing our understanding of carbohydrate biochemistry and the development of novel carbohydrate-based materials and methodologies.
Methyl 2-Acetamido-2-deoxy-β-D-galactopyranoside (CAS 3055-46-7) References
- Preparative production and separation of 2-acetamido-2-deoxymannopyranoside-containing saccharides using borate-saturated polyolic exclusion gels. | Petrásková, L., et al. 2006. J Chromatogr A. 1127: 126-36. PMID: 16814302
- Stereoselective entry into the D-GalNAc series starting from the D-Gal one: a new access to N-acetyl-D-galactosamine and derivatives thereof. | Guazzelli, L., et al. 2009. Carbohydr Res. 344: 298-303. PMID: 19128798
- Concise and efficient synthesis of 2-acetamido-2-deoxy-beta-D-hexopyranosides of diverse aminosugars from 2-acetamido-2-deoxy-beta-D-glucose. | Cai, Y., et al. 2009. J Org Chem. 74: 580-9. PMID: 19132941
- Explaining the Serological Characteristics of Streptococcus suis Serotypes 1 and 1/2 from Their Capsular Polysaccharide Structure and Biosynthesis. | Van Calsteren, MR., et al. 2016. J Biol Chem. 291: 8387-98. PMID: 26912653
- Unexpected Dissociation Mechanism of Sodiated N-Acetylglucosamine and N-Acetylgalactosamine. | Chiu, CC., et al. 2019. J Phys Chem A. 123: 3441-3453. PMID: 30945547
- Solution conformation of glycosaminoglycans: Assignment of the 300‐MHz 1H‐magnetic resonance spectra of chondroitin 4‐sulphate, chondroitin 6‐sulphate and hyaluronate, and investigation of an alkali‐induced conformation change. | Welti, et al. 1979. European Journal of Biochemistry. 94.2: 505-514.
- Diastereoselectivity in the transglycosidation of methyl 2-deoxy-3, 4, 6-tri-O-methyl-2 (N-methylacetamido)-d-glucopyranoside,-galactopyranoside, and-mannopyranoside with racemic 2-butanol under reductive-cleavage conditions. | Ahn, et al. 1997. Carbohydrate research. 298.4: 279-289.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Methyl 2-Acetamido-2-deoxy-β-D-galactopyranoside, 25 mg | sc-211823 | 25 mg | $300.00 |
