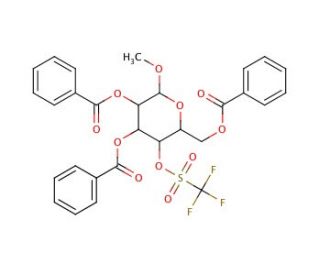
Methyl 2,3,6-Tri-O-benzoyl-4-O-trifluoromethanesulfononyl -α-D-galactopyranoside
QUICK LINKS
Methyl 2,3,6-Tri-O-benzoyl-4-O-trifluoromethanesulfononyl-α-D-galactopyranoside is a chemically sophisticated compound utilized primarily in the realm of synthetic organic chemistry and glycoscience. This molecule features a galactopyranoside core heavily modified with benzoyl groups at the 2, 3, and 6 positions and a trifluoromethanesulfononyl group at the 4 position. These modifications dramatically enhance the molecule′s stability and reactivity, making it an invaluable tool for studying complex glycosylation reactions. The trifluoromethanesulfononyl group, in particular, is a powerful leaving group that facilitates the formation of glycosidic bonds under mild conditions, making this compound an excellent glycosyl donor in the synthesis of complex oligosaccharides. The benzoyl protections prevent unwanted side reactions and help maintain the integrity of the glycosidic bond during synthetic manipulations. This derivative is crucial for exploring the stereoselectivity of glycosyltransferases and for developing new methodologies for assembling carbohydrates with precise structural configurations. Research utilizing this compound focuses on advancing the understanding of carbohydrate assembly processes, which are key to synthesizing biologically relevant glycoconjugates. Such studies are instrumental in pushing the boundaries of synthetic carbohydrate chemistry, providing insights that facilitate the development of new materials and biotechnological applications.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Methyl 2,3,6-Tri-O-benzoyl-4-O-trifluoromethanesulfononyl -α-D-galactopyranoside, 250 mg | sc-224070 | 250 mg | $360.00 |
