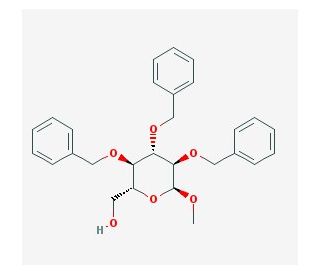
Methyl 2,3,4-tri-O-benzyl-α-D-glucopyranoside (CAS 53008-65-4)
QUICK LINKS
Methyl 2,3,4-tri-O-benzyl-α-D-glucopyranoside is a specialized chemical used primarily in synthetic organic chemistry, particularly in the synthesis of complex glycoconjugates. Its structure, featuring the benzyl protection of the 2, 3, and 4 hydroxyl groups of the glucose molecule, makes it an invaluable intermediate for the stepwise construction of oligosaccharides. The selective protection provided by the benzyl groups allows for specific reaction conditions where other unprotected hydroxyl groups can react, making it a crucial tool for controlling glycosylation reactions. This compound is often employed in the assembly of glycosidic linkages through pre-activation strategies followed by selective deprotection, enabling the synthesis of specific and structurally diverse glycostructures. Research utilizing this compound is primarily focused on understanding carbohydrate interactions at the molecular level, which are crucial for deciphering biological processes involving cell recognition and adhesion. Its use extends to the study of carbohydrate-based materials where precision in molecular architecture can lead to innovations in biomaterial science.
Methyl 2,3,4-tri-O-benzyl-α-D-glucopyranoside (CAS 53008-65-4) References
- Gold-catalyzed glycosidation for the synthesis of trisaccharides by applying the armed-disarmed strategy. | Kayastha, AK. and Hotha, S. 2013. Beilstein J Org Chem. 9: 2147-55. PMID: 24204427
- Stereocontrolled Synthesis of the Equatorial Glycosides of 3-Deoxy-d-manno-oct-2-ulosonic Acid: Role of Side Chain Conformation. | Ngoje, P. and Crich, D. 2020. J Am Chem Soc. 142: 7760-7764. PMID: 32275429
- A General Approach to O-Sulfation by a Sulfur(VI) Fluoride Exchange Reaction. | Liu, C., et al. 2020. Angew Chem Int Ed Engl. 59: 18435-18441. PMID: 32644280
- Addressing the Biochemical Foundations of a Glucose-Based 'Trojan Horse'-Strategy to Boron Neutron Capture Therapy: From Chemical Synthesis to In Vitro Assessment. | Matović, J., et al. 2020. Mol Pharm. 17: 3885-3899. PMID: 32787269
- Influence of substitution at the 5α-Position on the side chain conformation of glucopyranosides. | Rajasekaran, P., et al. 2021. Carbohydr Res. 500: 108254. PMID: 33561715
- Palladium(II)-assisted activation of thioglycosides. | Escopy, S., et al. 2021. Org Biomol Chem. 19: 2044-2054. PMID: 33599667
- Bismuth(iii) triflate as a novel and efficient activator for glycosyl halides. | Steber, HB., et al. 2021. Org Biomol Chem. 19: 3220-3233. PMID: 33885577
- Side Chain Conformation and Its Influence on Glycosylation Selectivity in Hexo- and Higher Carbon Furanosides. | Siyabalapitiya Arachchige, S. and Crich, D. 2022. J Org Chem. 87: 316-339. PMID: 34905382
- Efficient Synthesis of 2-OH Thioglycosides from Glycals Based on the Reduction of Aryl Disulfides by NaBH4. | Guo, YF., et al. 2022. Molecules. 27: PMID: 36144712
- Activation of Thioglycosides with Copper(II) Bromide. | Pooladian, F., et al. 2022. Molecules. 27: PMID: 36364179
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Methyl 2,3,4-tri-O-benzyl-α-D-glucopyranoside, 1 g | sc-235702 | 1 g | $151.00 |
