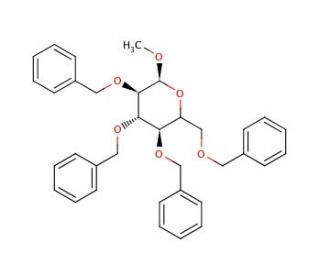
Methyl 2,3,4,6-Tetra-O-benzyl-α-D-mannopyranoside (CAS 61330-62-9)
QUICK LINKS
Methyl 2,3,4,6-Tetra-O-benzyl-α-D-mannopyranoside is a synthetically modified sugar extensively used in carbohydrate chemistry for its protective benzyl groups, which shield the hydroxyl groups during chemical synthesis, allowing for selective reactions at these sites. This compound is crucial in the preparation of complex glycosides and oligosaccharides, where precise control over glycosidic linkages is required. The methoxy group at the anomeric position serves as a stable protecting group that can be selectively removed or modified, facilitating subsequent glycosylation reactions. In research applications, this chemical′s ability to participate in the formation of glycosidic bonds under controlled conditions is particularly valuable in the synthesis of diverse and structurally complex carbohydrates. It plays a key role in studying the mechanisms of enzymatic glycosylation and deglycosylation, which are fundamental processes in cellular biology. This sugar derivative is also instrumental in the development of new materials and biomolecules in material science, where glycoengineering techniques are used to create functional materials with specific biological properties. Additionally, its application extends to the field of synthetic biology, where it aids in the construction of artificial biosynthetic pathways. Through these applications, Methyl 2,3,4,6-Tetra-O-benzyl-α-D-mannopyranoside contributes to advancements in understanding the chemical underpinnings of biological functions and the development of novel biochemical tools and materials.
Methyl 2,3,4,6-Tetra-O-benzyl-α-D-mannopyranoside (CAS 61330-62-9) References
- Synthesis of 3-C-(6-O-acetyl-2,3,4-tri-O-benzyl-alpha-D-mannopyranosyl)-1-propene: a caveat. | Girard, C., et al. 2002. Carbohydr Res. 337: 1769-74. PMID: 12423956
- Stereoselective synthesis of allyl-C-mannosyl compounds: use of a temporary silicon connection in intramolecular allylation strategies with allylsilanes. | Beignet, J., et al. 2004. J Org Chem. 69: 6341-56. PMID: 15357594
- Facile oxidative cleavage of 4-O-benzyl ethers with dichlorodicyanoquinone in rhamno- and mannopyranosides. | Crich, D. and Vinogradova, O. 2007. J Org Chem. 72: 3581-4. PMID: 17375956
- Microwave-assisted synthesis of novel purine nucleosides as selective cholinesterase inhibitors. | Schwarz, S., et al. 2014. Org Biomol Chem. 12: 2446-56. PMID: 24604285
- Nonhydrolyzable C-disaccharides, a new class of DC-SIGN ligands. | Bertolotti, B., et al. 2016. Carbohydr Res. 435: 7-18. PMID: 27676269
- C-Mannosyl Lysine for Solid Phase Assembly of Mannosylated Peptide Conjugate Cancer Vaccines. | Hogervorst, TP., et al. 2020. ACS Chem Biol. 15: 728-739. PMID: 32045202
- Maltoheptaose-Presenting Nanoscale Glycoliposomes for the Delivery of Rifampicin to E. coli. | Wu, B., et al. 2021. ACS Appl Nano Mater. 4: 7343-7357. PMID: 34746649
- Clarification of the Transfer Reaction in the Synthesis of Manno-oligosaccharide | Ota, N., & Hatanaka, K. 1998. Macromolecules. 31(7): 2123-2128.
- A one-pot selective deprotective acetylation of benzyl ethers and OTBDMS ethers using the BF3·Et2O–NaI–Ac2O reagent system | Brar, A., & Vankar, Y. D. 2006. Tetrahedron letters. 47(29): 5207-5210.
- New Method for Regioselective Glycosylation Employing Saccharide Oxyanions | Matwiejuk, M., & Thiem, J. 2011. EurJOC (European Journal of Organic Chemistry). 2011(29): 5860-5878.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Methyl 2,3,4,6-Tetra-O-benzyl-α-D-mannopyranoside, 2.5 g | sc-221906 | 2.5 g | $300.00 |
