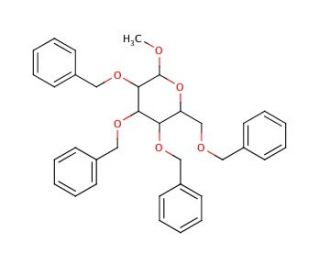
Methyl 2,3,4,6-Tetra-O-benzyl-α-D-glucopyranoside (CAS 17791-37-6)
QUICK LINKS
Methyl 2,3,4,6-Tetra-O-benzyl-α-D-glucopyranoside is a chemically modified sugar extensively used in carbohydrate chemistry for the study of glycosylation reactions, which are central to the synthesis of complex oligosaccharides and glycoconjugates. This compound features a glucose molecule where all the hydroxyl groups are protected by benzyl groups, making it a stable and reactive intermediate for synthesizing glycosidic bonds in a controlled manner. The presence of the methyl group at the anomeric position makes this derivative an excellent glycosyl donor in glycosylation reactions, where it can be used to transfer the glucose unit to other sugar acceptors under the action of catalysts, typically acids or enzymes. The benzyl protections on the hydroxyl groups prevent unwanted side reactions and can be selectively removed after glycosylation, allowing for subsequent elongation of the sugar chain. This specific functionality makes it invaluable for researchers aiming to construct accurately defined glycan structures that are essential for understanding carbohydrate biology. In synthetic organic chemistry, the use of such compounds helps explain mechanisms of bond formation and breakage, advancing the field of synthetic methodology and providing insights into the complex chemistry of carbohydrates.
Methyl 2,3,4,6-Tetra-O-benzyl-α-D-glucopyranoside (CAS 17791-37-6) References
- An expedient synthesis of benzyl 2,3,4-tri-O-benzyl-beta-D-glucopyranoside and benzyl 2,3,4-tri-O-benzyl-beta-D-mannopyranoside. | Lu, W., et al. 2005. Carbohydr Res. 340: 1213-7. PMID: 15797138
- Facile and regioselective preparation of partly O-benzylated D-glucopyranose acetates via acid-mediated simultaneous debenzylation-acetolysis. | Cao, Y., et al. 2006. Carbohydr Res. 341: 2219-23. PMID: 16806130
- Synthesis of septanosides through an oxyglycal route. | Ganesh, NV. and Jayaraman, N. 2007. J Org Chem. 72: 5500-4. PMID: 17585814
- Preparation of partially benzylated mono-, di-, and trisaccharides by selective cleavage of the beta-fructofuranosidic linkage in fully benzylated sucrose and sucrose-related oligosaccharides under acidic conditions. | Yamanoi, T., et al. 2008. Carbohydr Res. 343: 1366-72. PMID: 18423585
- Synthesis of the steroidal glycoside (25R)-3β,16β-diacetoxy-12,22-dioxo-5α-cholestan-26-yl β-D-glucopyranoside and its anti-cancer properties on cervicouterine HeLa, CaSki, and ViBo cells. | Fernández-Herrera, MA., et al. 2010. Eur J Med Chem. 45: 4827-37. PMID: 20801554
- Structure-activity relationships in a series of C2-substituted gluco-configured tetrahydroimidazopyridines as β-glucosidase inhibitors. | Li, T., et al. 2011. Bioorg Med Chem. 19: 2136-44. PMID: 21420868
- An α-selective, visible light photocatalytic glycosylation of alcohols with selenoglycosides. | Spell, M., et al. 2013. Carbohydr Res. 369: 42-7. PMID: 23399745
- Streamlined access to carbohydrate building blocks: Methyl 2,4,6-tri-O-benzyl-α-d-glucopyranoside. | Shrestha, G., et al. 2022. Carbohydr Res. 511: 108482. PMID: 34856429
- Debenzylation of carbohydrate benzyl ethers and benzyl glycosides via free-radical bromination | BeMiller, J. N., & Muenchow, H. L. 1973. Carbohydrate Research. 28(2): 253-262.
- The allyl ether as a protecting group in carbohydrate chemistry. Part V. Preparation of benzyl ethers of carbohydrates for use in oligosaccharide synthesis | Gent, P. A., & Gigg, R. 1974. Journal of the Chemical Society, Perkin Transactions 1. 0: 1446-1455.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Methyl 2,3,4,6-Tetra-O-benzyl-α-D-glucopyranoside, 5 g | sc-221905 | 5 g | $330.00 |
