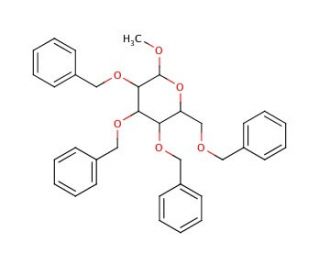
Methyl 2,3,4,6-Tetra-O-benzyl-α-D-galactopyranoside (CAS 53008-63-2)
QUICK LINKS
Methyl 2,3,4,6-Tetra-O-benzyl-α-D-galactopyranoside serves as a versatile tool in carbohydrate chemistry research, particularly in the synthesis of complex oligosaccharides and glycoconjugates. Its chemical structure, featuring a galactopyranoside core with four benzyl protecting groups, offers unique reactivity and selectivity in glycosylation reactions. Chemists utilize this compound as a key building block to introduce galactose residues into carbohydrate chains with precise control over regio- and stereochemistry. Through strategic manipulation of reaction conditions and protecting group strategies, researchers can access a wide range of structurally diverse glycosides and glycoconjugates. Additionally, Methyl 2,3,4,6-Tetra-O-benzyl-α-D-galactopyranoside serves as a valuable substrate for enzymatic glycosylation reactions, enabling the chemoenzymatic synthesis of complex carbohydrates and glycoconjugates with high efficiency and selectivity. Furthermore, this compound has found applications in the development of carbohydrate-based materials, such as glycosylated polymers and biomimetic surfaces, for various biotechnological and biomedical applications. Overall, Methyl 2,3,4,6-Tetra-O-benzyl-α-D-galactopyranoside plays a pivotal role in advancing research in carbohydrate chemistry, glycobiology, and related fields, offering valuable insights into carbohydrate structure-function relationships and enabling the design and synthesis of novel glycotherapeutics and biomaterials.
Methyl 2,3,4,6-Tetra-O-benzyl-α-D-galactopyranoside (CAS 53008-63-2) References
- Synthesis of cis-(1-->3)-glycosides of allyl 2-acetamido-4,6-O-benzylidene-2-deoxy-alpha-D-glucopyranoside. | Madaj, J., et al. 2004. Carbohydr Res. 339: 1293-300. PMID: 15113666
- Synthesis and biological activity of ester and ether analogues of alpha-galactosylceramide (KRN7000). | Shiozaki, M., et al. 2010. Carbohydr Res. 345: 1663-84. PMID: 20591421
- Total synthesis of α-1C-galactosylceramide, an immunostimulatory C-glycosphingolipid, and confirmation of the stereochemistry in the first-generation synthesis. | Liu, Z., et al. 2011. J Org Chem. 76: 8588-98. PMID: 21958232
- Synthesis and biological activity of hydroxylated analogues of KRN7000 (α-galactosylceramide). | Shiozaki, M., et al. 2013. Carbohydr Res. 370: 46-66. PMID: 23454137
- Microwave-assisted synthesis of novel purine nucleosides as selective cholinesterase inhibitors. | Schwarz, S., et al. 2014. Org Biomol Chem. 12: 2446-56. PMID: 24604285
- New antitumor 6-chloropurine nucleosides inducing apoptosis and G2/M cell cycle arrest. | Schwarz, S., et al. 2015. Eur J Med Chem. 90: 595-602. PMID: 25499928
- Use of positively charged, leaving groups in the synthesis of α-D-linked galactosides. Attempted synthesis of 3-O-α-(D-(galactopyranosyl)-D-galactose | Kronzer, F. J., & Schuerch, C. 1974. Carbohydrate Research. 33(2): 273-280.
- A one-pot selective deprotective acetylation of benzyl ethers and OTBDMS ethers using the BF3· Et2O–NaI–Ac2O reagent system | Brar, A., & Vankar, Y. D. 2006. Tetrahedron letters. 47(29): 5207-5210.
- New method for regioselective glycosylation employing saccharide oxyanion | Matwiejuk, M., & Thiem, J. 2011. EurJOC (European Journal of Organic Chemistry). 2011 (29): 5860-5878.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Methyl 2,3,4,6-Tetra-O-benzyl-α-D-galactopyranoside, 1 g | sc-221904 | 1 g | $300.00 |
