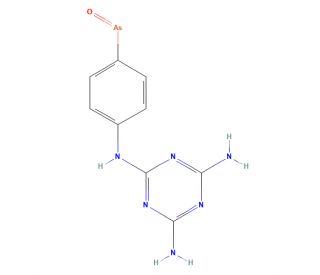
Molecular structure of Melarsen Oxide, CAS Number: 21840-08-4
Melarsen Oxide (CAS 21840-08-4)
Alternate Names:
N2-(4-Arsenosophenyl)-1,3,5-triazine-2,4,6-triamine; (p-Arsenosophenyl)melamine; (p-Arsenosophenyl)melamine
CAS Number:
21840-08-4
Molecular Weight:
292.13
Molecular Formula:
C9H9AsN6O
Supplemental Information:
This is classified as a Dangerous Good for transport and may be subject to additional shipping charges.
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Melarsen oxide, recognized as the dynamic metabolite resulting from the metabolic conversion of Melarsoprol, operates through a specific biochemical mechanism as a glutathione reductase inhibitor. This action is pivotal in the context of biochemical research, particularly in studies focusing on the cellular defense mechanisms against oxidative stress. By inhibiting glutathione reductase, Melarsen oxide disrupts the glutathione cycle, a pathway for maintaining cellular redox balance. This disruption leads to an accumulation of oxidative products within the cell, thereby offering a unique model for understanding the cellular responses to oxidative damage.
Melarsen Oxide (CAS 21840-08-4) References
- Investigations of the metabolites of the trypanocidal drug melarsoprol. | Keiser, J., et al. 2000. Clin Pharmacol Ther. 67: 478-88. PMID: 10824626
- Four years' experience of melarsen oxide/BAL in the treatment of late-stage Rhodesian sleeping sickness. | APTED, FI. 1957. Trans R Soc Trop Med Hyg. 51: 75-86. PMID: 13409596
- Relative efficacy of melarsen oxide compared with mel Cy (Cymelarsan) when used in combination with difluoromethylornithine in the treatment of trypanosomiasis of the central nervous system. | Jennings, FW. 1992. Trans R Soc Trop Med Hyg. 86: 257-8. PMID: 1412647
- The melaminophenyl arsenicals melarsoprol and melarsen oxide interfere with thiamine metabolism in the fission yeast Schizosaccharomyces pombe. | Schweingruber, ME. 2004. Antimicrob Agents Chemother. 48: 3268-71. PMID: 15328083
- Biosynthesis and uptake of thiamine (vitamin B1) in bloodstream form Trypanosoma brucei brucei and interference of the vitamin with melarsen oxide activity. | Stoffel, SA., et al. 2006. Int J Parasitol. 36: 229-36. PMID: 16375907
- Melarsen oxide in the treatment of human trypanosomiasis. | FRIEDHEIM, EA. 1948. Ann Trop Med Parasitol. 42: 357-63. PMID: 18110349
- Preliminary observations on the use of melarsen oxide in malaria. | PAYNE, EH., et al. 1946. South Med J. 39: 970-2. PMID: 20274941
- Further experience with melarsen oxide (a new arsenical) in the treatment of tropical disease. | PAYNE, EH., et al. 1946. South Med J. 39: 972-5. PMID: 20274942
- The treatment of African sleeping sickness with two new trivalent arsenical preparations (melarsen oxide and 70A). | WEINMAN, D. 1946. Am J Trop Med Hyg. 26: 95-105. PMID: 21003285
- Trypanothione is the primary target for arsenical drugs against African trypanosomes. | Fairlamb, AH., et al. 1989. Proc Natl Acad Sci U S A. 86: 2607-11. PMID: 2704738
- Effects of various metabolic conditions and of the trivalent arsenical melarsen oxide on the intracellular levels of fructose 2,6-bisphosphate and of glycolytic intermediates in Trypanosoma brucei. | Van Schaftingen, E., et al. 1987. Eur J Biochem. 166: 653-61. PMID: 3038548
- Differential susceptibility of filarial and human erythrocyte glutathione reductase to inhibition by the trivalent organic arsenical melarsen oxide. | Müller, S., et al. 1995. Mol Biochem Parasitol. 71: 211-9. PMID: 7477103
- Properties of melarsamine hydrochloride (Cymelarsan) in aqueous solution. | Berger, BJ. and Fairlamb, AH. 1994. Antimicrob Agents Chemother. 38: 1298-302. PMID: 8092828
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Melarsen Oxide, 50 mg | sc-488923 | 50 mg | $388.00 |
