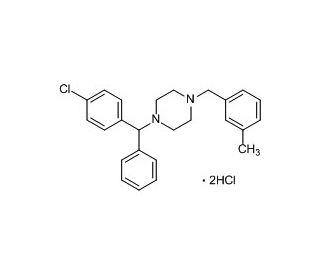
Molecular structure of Meclizine Dihydrochloride, CAS Number: 1104-22-9
Meclizine Dihydrochloride (CAS 1104-22-9)
See product citations (3)
Alternate Names:
Bonamine; Ancolan dihydrochloride; Antivert
Application:
Meclizine Dihydrochloride is a histamine H1 receptor antagonist and PXR (pregnane X receptor) agonist
CAS Number:
1104-22-9
Purity:
≥98%
Molecular Weight:
463.87
Molecular Formula:
C25H27ClN2•2HCl
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Product Citations
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Meclizine Dihydrochloride is a PXR (pregnane X receptor) agonist and histamine H1 receptor antagonist. The compound also possesses anticholinergic, central nervous system depressant, and local anesthetic effects. Meclizine depresses labyrinth excitability and vestibular stimulation and may affect the medullary chemoreceptor. Meclizine Dihydrochloride shares similarities with phenethylamine, but with a methoxy group positioned at the 4th spot.
Meclizine Dihydrochloride (CAS 1104-22-9) References
- The major role of peripheral release of histamine and 5-hydroxytryptamine in formalin-induced nociception. | Parada, CA., et al. 2001. Neuroscience. 102: 937-44. PMID: 11182255
- Quantitative enantiomeric analysis of chlorcyclizine, hydroxyzine, and meclizine by capillary electrophoresis. | Ho, Y., et al. 2003. Anal Bioanal Chem. 376: 859-63. PMID: 12830360
- Determination of flunarizine in plasma by a new high-performance liquid chromatography method. Application to a bioavailability study in the rat. | Aparicio, X., et al. 1988. J Pharm Biomed Anal. 6: 167-73. PMID: 16867429
- Meclizine is neuroprotective in models of Huntington's disease. | Gohil, VM., et al. 2011. Hum Mol Genet. 20: 294-300. PMID: 20977989
- Meclizine inhibits mitochondrial respiration through direct targeting of cytosolic phosphoethanolamine metabolism. | Gohil, VM., et al. 2013. J Biol Chem. 288: 35387-95. PMID: 24142790
- Cisplatin Toxicity in Dorsal Root Ganglion Neurons Is Relieved by Meclizine via Diminution of Mitochondrial Compromise and Improved Clearance of DNA Damage. | Gorgun, MF., et al. 2017. Mol Neurobiol. 54: 7883-7895. PMID: 27858292
- Suncus murinus as a new experimental model for motion sickness. | Ueno, S., et al. 1988. Life Sci. 43: 413-20. PMID: 2899827
- Cascade Ligand- and Structure-Based Virtual Screening to Identify New Trypanocidal Compounds Inhibiting Putrescine Uptake. | Alberca, LN., et al. 2018. Front Cell Infect Microbiol. 8: 173. PMID: 29888213
- Metal-free photocatalysts for the oxidation of non-activated alcohols and the oxygenation of tertiary amines performed in air or oxygen. | Zhang, Y., et al. 2020. Nat Protoc. 15: 822-839. PMID: 32051614
- Prenatal ethanol exposure increases maternal bile acids through placental transport pathway. | Liu, F., et al. 2021. Toxicology. 458: 152848. PMID: 34217791
- Comparison of meclizine levels in the plasma of rats and dogs after intranasal, intravenous, and oral administration. | Chovan, JP., et al. 1985. J Pharm Sci. 74: 1111-3. PMID: 4078711
- pKa determination of benzhydrylpiperazine antihistamines in aqueous and aqueous methanol solutions. | Newton, DW., et al. 1982. J Pharm Sci. 71: 1363-6. PMID: 6130141
- Comparison of the effects of eleven histamine H1-receptor antagonists on monoamine turnover in the mouse brain. | Oishi, R., et al. 1994. Naunyn Schmiedebergs Arch Pharmacol. 349: 140-4. PMID: 7513381
- Pharmaceutical properties of freeze-dried formulations of egg albumin, several drugs and olive oil. | Tsuji, Y., et al. 1996. Biol Pharm Bull. 19: 636-40. PMID: 9132174
Inhibitor of:
Histamine H1 Receptor.Activator of:
PXR.Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Meclizine Dihydrochloride, 5 g | sc-211779A | 5 g | $67.00 | |||
Meclizine Dihydrochloride, 10 g | sc-211779 | 10 g | $141.00 |
