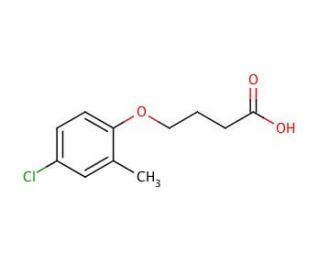
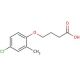
MCPB (CAS 94-81-5)
QUICK LINKS
The versatility of MCPB extends to its involvement in various scientific research applications. Furthermore, its utilization extends to the creation of polymers, plastics, and other materials employed in diverse industries. This organic compound also plays a role in the synthesis of polyacetylenes and polyamides, key components in the production of plastics and other polymeric materials. While the precise mechanism of action for MCPB remains not fully elucidated, it is postulated to function as a proton donor. By donating a proton to a substrate molecule, it facilitates the progression of a reaction. Additionally, this compound is believed to exhibit catalyst properties, thereby promoting the formation of new products from multiple reactants.
MCPB (CAS 94-81-5) References
- Solid-phase extraction of acidic herbicides. | Wells, MJ. and Yu, LZ. 2000. J Chromatogr A. 885: 237-50. PMID: 10941675
- Characterisation of bacterial cultures enriched on the chlorophenoxyalkanoic acid herbicides 4-(2,4-dichlorophenoxy) butyric acid and 4-(4-chloro-2-methylphenoxy) butyric acid. | Smejkal, CW., et al. 2003. J Ind Microbiol Biotechnol. 30: 561-7. PMID: 14513383
- Capillary liquid chromatography of chlorophenoxy acid herbicides and their esters in apple juice samples after preconcentration on a cation exchanger based on polydivinylbenzene-N-vinylpyrrolidone. | Rosales-Conrado, N., et al. 2005. J Chromatogr A. 1076: 202-6. PMID: 15974090
- Multiresidue determination of chlorophenoxy acid herbicides in human urine samples by use of solid-phase extraction and capillary LC-UV detection. | Rosales-Conrado, N., et al. 2008. Anal Bioanal Chem. 390: 759-68. PMID: 17987282
- Selective inhibition of histone deacetylases sensitizes malignant cells to death receptor ligands. | Wood, TE., et al. 2010. Mol Cancer Ther. 9: 246-56. PMID: 20053768
- Development of hapten-linked microimmunoassays on polycarbonate discs. | Tamarit-López, J., et al. 2010. Anal Chem. 82: 1954-63. PMID: 20131798
- Screening method for phenoxy acid herbicides in ground water by high-performance liquid chromatography of 9-anthryldiazomethane derivatives and fluorescence detection. | Suzuki, T. and Watanabe, S. 1991. J Chromatogr. 541: 359-64. PMID: 2037653
- Comparative analysis of QSAR models for predicting pK(a) of organic oxygen acids and nitrogen bases from molecular structure. | Yu, H., et al. 2010. J Chem Inf Model. 50: 1949-60. PMID: 21033677
- Oxidative degradation of different chlorinated phenoxyalkanoic acid herbicides by a hybrid ZrO2 gel-derived catalyst without light irradiation. | Sannino, F., et al. 2015. ACS Appl Mater Interfaces. 7: 256-63. PMID: 25479367
- Adsorption and degradation of phenoxyalkanoic acid herbicides in soils: A review. | Paszko, T., et al. 2016. Environ Toxicol Chem. 35: 271-86. PMID: 26292078
- Selective and Sensitive Chemiluminescence Determination of MCPB: Flow Injection and Liquid Chromatography. | Meseguer-Lloret, S., et al. 2016. Appl Spectrosc. 70: 312-21. PMID: 26903566
- Response of the bacterial community in an on-farm biopurification system, to which diverse pesticides are introduced over an agricultural season. | Holmsgaard, PN., et al. 2017. Environ Pollut. 229: 854-862. PMID: 28734695
- A review of recent developments based on chemiluminescence detection systems for pesticides analysis. | Al Yahyai, I. and Al-Lawati, HAJ. 2021. Luminescence. 36: 266-277. PMID: 32909300
- Magnetic molecularly imprinted polymer for the simultaneous selective extraction of phenoxy acid herbicides from environmental water samples. | Meseguer-Lloret, S., et al. 2022. Talanta. 239: 123082. PMID: 34823860
- Etherolytic cleavage of 4-(2,4-dichlorophenoxy)butyric acid and 4-(4-chloro-2-methylphenoxy)butyric acid by species of Rhodococcus and Aureobacterium isolated from an alkaline environment. | Mertingk, H., et al. 1998. J Basic Microbiol. 38: 257-267. PMID: 9867509
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
MCPB, 100 mg | sc-250295 | 100 mg | $79.00 |
