MC 1293
See product citations (1)
QUICK LINKS
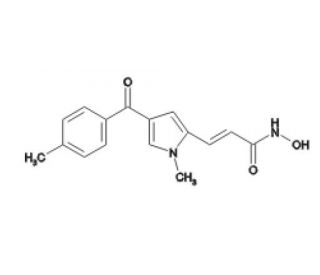
MC 1293 is a chemical compound known for its role as an inhibitor of histone deacetylase 1 (HDAC1) and maize histone deacetylase HD2. Its inhibition capabilities are quantified with IC50 values of 4.5µM for HDAC1 and 1.9µM for maize HD2, indicating its potency at these specific concentrations. MC 1293 is particularly valuable for studying the modulation of chromatin structure through epigenetic mechanisms. HDAC inhibitors like MC 1293 are used to explore how changes in histone acetylation affect the organization and accessibility of chromatin, thereby influencing cellular function and gene expression. This area of research is crucial for understanding the fundamental processes that govern cellular regulation and the maintenance of genomic integrity. By targeting specific HDAC enzymes, MC 1293 serves as a useful experimental tool in molecular biology and biochemistry for examining the impact of histone acetylation on various cellular processes. The specificity and effectiveness of MC 1293 in inhibiting particular HDACs highlight its importance in the toolkit for research in epigenetic regulation.
MC 1293 References
- 3-(4-aroyl-1H-pyrrol-2-yl)-N-hydroxy-2-propenamides, a new class of synthetic histone deacetylase inhibitors. | Massa, S., et al. 2001. J Med Chem. 44: 2069-72. PMID: 11405644
- Binding mode analysis of 3-(4-benzoyl-1-methyl-1H-2-pyrrolyl)-N-hydroxy-2-propenamide: a new synthetic histone deacetylase inhibitor inducing histone hyperacetylation, growth inhibition, and terminal cell differentiation. | Mai, A., et al. 2002. J Med Chem. 45: 1778-84. PMID: 11960489
- Inhibition of iNOS expression and NO production by anti-inflammatory steroids. Reversal by histone deacetylase inhibitors. | Hämäläinen, M., et al. 2008. Pulm Pharmacol Ther. 21: 331-9. PMID: 17913526
- Immunomodulatory effects of deacetylase inhibitors: therapeutic targeting of FOXP3+ regulatory T cells. | Wang, L., et al. 2009. Nat Rev Drug Discov. 8: 969-81. PMID: 19855427
- Histone deacetylase inhibitors induce apoptosis in human eosinophils and neutrophils. | Kankaanranta, H., et al. 2010. J Inflamm (Lond). 7: 9. PMID: 20181093
- A substrate-independent TR-FRET histone deacetylase inhibitor assay. | Marks, BD., et al. 2011. J Biomol Screen. 16: 1247-53. PMID: 21940713
- Histone deacetylase inhibitor MC1293 induces latent HIV-1 reactivation by histone modification in vitro latency cell lines. | Qu, X., et al. 2013. Curr HIV Res. 11: 24-9. PMID: 23092175
- Epigenetically maintained SW13+ and SW13- subtypes have different oncogenic potential and convert with HDAC1 inhibition. | Davis, MR., et al. 2016. BMC Cancer. 16: 316. PMID: 27188282
- The Distinct Roles of Class I and II RPD3-Like Histone Deacetylases in Salinity Stress Response. | Ueda, M., et al. 2017. Plant Physiol. 175: 1760-1773. PMID: 29018096
- LIM-Domain Protein Ajuba Is a Required Co-Factor for Gfi1-Induced Epigenetic Switch Regulating Runx2 Repression in Multiple Myeloma-Exposed Pre-Osteoblasts | Adamik, J., et al. 2015. Blood. 126(23): 4216.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
MC 1293, 5 mg | sc-221875 | 5 mg | $202.00 |
