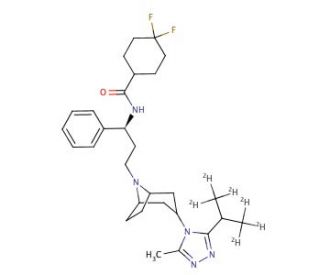
Maraviroc-d6
See product citations (4)
QUICK LINKS
Maraviroc-d6 is a deuterated form of maraviroc, where six hydrogen atoms are replaced with deuterium. This modification is often used in scientific research to enhance the accuracy and sensitivity of various analytical methods, particularly in the study of the compound′s behavior in biological systems. Maraviroc is known for its role as a CCR5 antagonist. It binds selectively to the CCR5 receptor on the surface of human cells, which is a co-receptor used by certain strains of HIV to enter and infect host cells. By blocking this receptor, maraviroc-d6 prevents the virus from attaching to and entering the host cells, thereby inhibiting viral replication. This mechanism of action remains unchanged in the deuterated form, maraviroc-d6, allowing researchers to study the interaction with the CCR5 receptor in great detail. In research, maraviroc-d6 is particularly useful for tracing the pharmacokinetics and dynamics of maraviroc without the interference that might affect non-deuterated forms. The deuterated atoms improve the detection and quantitation in mass spectrometry, making it easier to monitor how the compound is absorbed, distributed, metabolized, and excreted in biological systems. Furthermore, maraviroc-d6 is used in studies focusing on the metabolic pathways of maraviroc. Researchers use this deuterated version to identify and characterize the metabolic enzymes responsible for processing the compound. This helps in understanding how genetic and environmental factors might influence the metabolism of maraviroc and similar molecules.
Maraviroc-d6 References
- Cervicovaginal and Rectal Fluid as a Surrogate Marker of Antiretroviral Tissue Concentration: Implications for Clinical Trial Design. | Cottrell, ML., et al. 2016. J Acquir Immune Defic Syndr. 72: 498-506. PMID: 26999532
- Brief Report: Pharmacokinetic/Pharmacodynamic Investigation of Single-Dose Oral Maraviroc in the Context of HIV-1 Pre-exposure Prophylaxis. | Fox, J., et al. 2016. J Acquir Immune Defic Syndr. 73: 252-257. PMID: 27727157
- Simultaneous determination of cytosine arabinoside and its metabolite uracil arabinoside in human plasma by LC-MS/MS: Application to pharmacokinetics-pharmacogenetics pilot study in AML patients. | Donnette, M., et al. 2019. J Chromatogr B Analyt Technol Biomed Life Sci. 1126-1127: 121770. PMID: 31454720
- Pharmacokinetics of a weekly transdermal delivery system of tenofovir alafenamide in hairless rats. | Jiang, Y., et al. 2020. Int J Pharm. 582: 119342. PMID: 32315746
- Interplay of drug transporters P-glycoprotein (MDR1), MRP1, OATP1A2 and OATP1B3 in passage of maraviroc across human placenta. | Tupova, L., et al. 2020. Biomed Pharmacother. 129: 110506. PMID: 32768979
- The ex vivo pharmacology of HIV-1 antiretrovirals differs between macaques and humans. | Herrera, C., et al. 2022. iScience. 25: 104409. PMID: 35663021
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Maraviroc-d6, 1 mg | sc-218671 | 1 mg | $430.00 |
