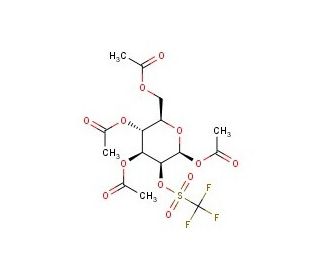
Mannose Triflate (CAS 92051-23-5)
QUICK LINKS
Mannose triflate, chemically designated as a triflate ester of mannose, is employed primarily in synthetic organic chemistry and complex carbohydrate synthesis research. Its utility derives from its role as a glycosyl donor in glycosylation reactions, which are fundamental for constructing oligosaccharide chains. This chemical is particularly valuable due to its high reactivity, which is imparted by the triflate group, making it an effective agent in promoting glycosidic bond formation under milder reaction conditions compared to traditional methods. Mannose triflate is critical in the study of carbohydrate-based interactions at the molecular level, such as those involving lectins, which are proteins that recognize specific carbohydrate moieties. Researchers utilize this compound to synthesize glycoconjugates, which are crucial for exploring biological recognition processes and cell signaling mechanisms. Its application extends to the field of materials science as well, where glyco-functionalized surfaces are studied for their biocompatibility and sensor applications. The role of mannose triflate in these research settings underscores its importance in advancing our understanding of biochemical pathways and molecular interactions.
Mannose Triflate (CAS 92051-23-5) References
- Chemical impurities in [18F]FDG preparations produced by solid-phase 18F-fluorination. | Kuge, Y., et al. 2002. Nucl Med Biol. 29: 275-9. PMID: 11823133
- Ion chromatographic analysis of high specific activity 18FDG preparations and detection of the chemical impurity 2-deoxy-2-chloro-D-glucose. | Alexoff, DL., et al. 1992. Int J Rad Appl Instrum A. 43: 1313-22. PMID: 1333458
- Practical and reliable synthesis of 1,3,4,6-tetra-O-acetyl-2-O-trifluoromethanesulfonyl-beta-D-mannopyranose, a precursor of 2-deoxy-2-[18F]fluoro-D-glucose (FDG). | Toyokuni, T., et al. 2004. Mol Imaging Biol. 6: 324-30. PMID: 15380742
- Microfluidic technology for PET radiochemistry. | Gillies, JM., et al. 2006. Appl Radiat Isot. 64: 333-6. PMID: 16290947
- Synthesis of the positron-emitting radiotracer [(18)F]-2-fluoro-2-deoxy-D-glucose from resin-bound perfluoroalkylsulfonates. | Brown, LJ., et al. 2009. Org Biomol Chem. 7: 564-75. PMID: 19156324
- Nucleophilic radiosynthesis of 2-[18F]fluoro-2-deoxy-D-galactose from Talose triflate and biodistribution in a porcine model. | Frisch, K., et al. 2011. Nucl Med Biol. 38: 477-83. PMID: 21531284
- Review of F-FDG Synthesis and Quality Control. | Yu, S. 2006. Biomed Imaging Interv J. 2: e57. PMID: 21614337
- Gold nanoparticle probes: design and in vitro applications in cancer cell culture. | Unak, G., et al. 2012. Colloids Surf B Biointerfaces. 90: 217-26. PMID: 22070896
- Automated solid-phase radiofluorination using polymer-supported phosphazenes. | Mathiessen, B. and Zhuravlev, F. 2013. Molecules. 18: 10531-47. PMID: 23999726
- Sweetening pharmaceutical radiochemistry by (18)f-fluoroglycosylation: a short review. | Maschauer, S. and Prante, O. 2014. Biomed Res Int. 2014: 214748. PMID: 24991541
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Mannose Triflate, 100 mg | sc-221870 | 100 mg | $230.00 |
