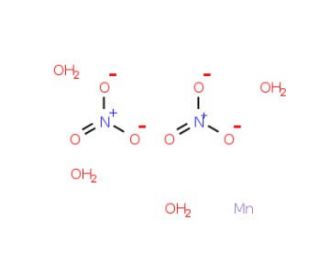
Molecular structure of Manganese(II) nitrate tetrahydrate, CAS Number: 20694-39-7
Manganese(II) nitrate tetrahydrate (CAS 20694-39-7)
CAS Number:
20694-39-7
Molecular Weight:
251.01 (178.95anhy)
Molecular Formula:
MnN2O6•4H2O
Supplemental Information:
This is classified as a Dangerous Good for transport and may be subject to additional shipping charges.
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Manganese(II) nitrate tetrahydrate is a hydrated manganese salt with water solubility, finding diverse applications such as compound synthesis, catalysis, and laboratory experimentation. In biochemical studies, it serves as a manganese source. Moreover, it plays a role in investigating plant physiology, particularly in assessing manganese uptake by plants.
Manganese(II) nitrate tetrahydrate (CAS 20694-39-7) References
- Effect of Heat Treatment on Structural, Magnetic and Electrical Properties of La2FeMnO6. | Triyono, D., et al. 2021. Materials (Basel). 14: PMID: 34947097
- Stability of Manganese(II)-Pyrazine, -Quinoxaline or -Phenazine Complexes and Their Potential as Carbonate Sequestration Agents. | Segoviano-Garfias, JJN., et al. 2022. Molecules. 27: PMID: 35268748
- Role of Bimetallic Solutions in the Growth and Functionality of Cu-BTC Metal-Organic Framework. | Gupta, NK., et al. 2022. Materials (Basel). 15: PMID: 35454498
- Synthesis, structural and luminescent properties of Mn-doped calcium pyrophosphate (Ca2P2O7) polymorphs. | Griesiute, D., et al. 2022. Sci Rep. 12: 7116. PMID: 35504944
- Structurally distorted perovskite La0.8Sr0.2Mn0.5Co0.5O3-δ by graphene nanoplatelet and their composite for supercapacitors with enhanced stability. | Kim, BM., et al. 2022. Sci Rep. 12: 10043. PMID: 35710937
- Development of High-Efficiency, Magnetically Separable Palladium-Decorated Manganese-Ferrite Catalyst for Nitrobenzene Hydrogenation. | Hajdu, V., et al. 2022. Int J Mol Sci. 23: PMID: 35742977
- Ternary metal oxide nanocomposite for room temperature H2S and SO2 gas removal in wet conditions. | Gupta, NK., et al. 2022. Sci Rep. 12: 15387. PMID: 36100623
- Spin-Crossover and Slow Magnetic Relaxation Behavior in Hexachlororhenate(IV) Salts of Mn(III) Complexes [Mn(5-Hal-sal2323)]2[ReCl6] (Hal = Cl, Br). | Tiunova, AV., et al. 2022. Int J Mol Sci. 23: PMID: 36232753
- Effect of Al and Fe Doping on the Electrochemical Behavior of Li1.2Ni0.133Mn0.534Co0.133O2 Li-Rich Cathode Material. | Medvedeva, A., et al. 2022. Materials (Basel). 15: PMID: 36431711
- Sustainable Removal of BTEX Gas Using Regenerated Metal Containing SiO2. | Beak, S., et al. 2022. Nanomaterials (Basel). 12: PMID: 36500736
- Enhancing catalytic efficiency of carbon dots by modulating their Mn doping and chemical structure with metal salts. | Kang, W., et al. 2023. RSC Adv. 13: 8996-9002. PMID: 36936848
- Mixed Transition-Metal Oxides on Reduced Graphene Oxide as a Selective Catalyst for Alkaline Oxygen Reduction. | Wolf, S., et al. 2023. ACS Omega. 8: 11536-11543. PMID: 37008156
- Temperature-Induced Irreversible Structural Transition in Fe1.1Mn1.9O4 Nanoparticles Synthesized by Combustion Method. | Spivakov, AA., et al. 2023. Nanomaterials (Basel). 13: PMID: 37049366
- A glucose-assisted redox hydrothermal route to prepare a Mn-doped CeO2 catalyst for the total catalytic oxidation of VOCs. | Phan, NHT., et al. 2023. RSC Adv. 13: 13354-13364. PMID: 37143917
- Preparation, performance and mechanism of metal oxide modified catalytic ceramic membranes for wastewater treatment. | Huang, Y., et al. 2023. RSC Adv. 13: 17436-17448. PMID: 37313519
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Manganese(II) nitrate tetrahydrate, 250 g | sc-269377 | 250 g | $84.00 |
