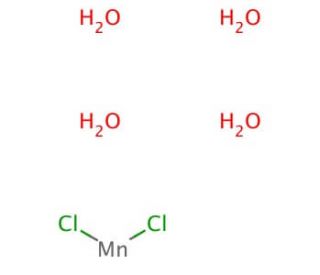
Manganese Chloride Tetrahydrate (CAS 13446-34-9)
QUICK LINKS
Manganese chloride tetrahydrate (MnCl2·4H2O) is an inorganic compound composed of manganese, chlorine, and water. It presents itself as a white, odorless powder that readily dissolves in water and exhibits slight solubility in alcohol. Widely employed as a laboratory reagent, it finds versatile applications in research, encompassing organic compound synthesis, biochemical analysis, and catalytic roles in chemical reactions. It has proven effective as a catalyst in synthesizing a range of organic compounds, including aldehydes, ketones, and carboxylic acids. Furthermore, it has found utility in the production of polymers and other materials. The compound functions as an oxidizing agent in both organic synthesis and biochemical analysis. In organic synthesis, it facilitates the oxidation of organic compounds, leading to the desired product formation. In the context of biochemical analysis, manganese chloride tetrahydrate serves as a reagent for the detection of various compounds, including amino acids, carbohydrates, proteins, and metals such as iron, copper, and zinc. It oxidizes the compounds under investigation, enabling their detection and quantification. Notably, manganese chloride tetrahydrate serves as useful in detecting phospholipid vesicles. In summary, manganese chloride tetrahydrate is a widely utilized inorganic compound, that acts as an oxidizing agent in facilitating organic reactions and biochemical analysis.
Manganese Chloride Tetrahydrate (CAS 13446-34-9) References
- Effects of long-term exposure to manganese chloride on fertility of male and female mice. | Elbetieha, A., et al. 2001. Toxicol Lett. 119: 193-201. PMID: 11246172
- Effects of prenatal exposure to manganese on postnatal development and behavior in mice: influence of maternal restraint. | Torrente, M., et al. 2002. Neurotoxicol Teratol. 24: 219-25. PMID: 11943509
- Abnormal motor function and the expression of striatal dopamine D2 receptors in manganese-treated mice. | Nam, J. and Kim, K. 2008. Biol Pharm Bull. 31: 1894-7. PMID: 18827350
- Synthesis, structural investigation and thermal properties of a novel manganese complex Mn2(DAT)2Cl4(H2O)4 (DAT=1,5-diaminotetrazole). | Zhang, JG., et al. 2010. J Hazard Mater. 178: 1094-9. PMID: 20338686
- A new manganese-based oral contrast agent (CMC-001) for liver MRI: pharmacological and pharmaceutical aspects. | Jørgensen, JT., et al. 2012. Acta Radiol. 53: 707-13. PMID: 22821959
- MR signal changes on hepatobiliary imaging after oral ingestion of manganese chloride tetrahydrate: preliminary examination. | Marugami, N., et al. 2013. Jpn J Radiol. 31: 713-23. PMID: 24068585
- Synthesis of manganese nanoparticles in the liquid phase plasma. | Kim, HG., et al. 2013. J Nanosci Nanotechnol. 13: 6103-8. PMID: 24205609
- [A Low-temperature Manganese Chloride Tetrahydrate Improves the Image Quality of Magnetic Resonance Cholangiopancreatography]. | Watanabe, K., et al. 2016. Nihon Hoshasen Gijutsu Gakkai Zasshi. 72: 311-8. PMID: 27097992
- Influence on Uranium(VI) migration in soil by iron and manganese salts of humic acid: Mechanism and behavior. | Zhang, YY., et al. 2020. Environ Pollut. 256: 113369. PMID: 31662254
- Relative efficacy of chelating agents on excretion and tissue distribution of manganese in mice. | Sánchez, DJ., et al. 1995. J Appl Toxicol. 15: 285-8. PMID: 7594197
- Manganese chloride as an efficient catalyst for selective transformations of indoles in the presence of a keto carbonyl group | Li, M., Yang, J., & Gu, Y. (2011). 2011. Advanced Synthesis & Catalysis. 353(9): 1551-1564.
- Interactions of some amino acids with aqueous manganese chloride tetrahydrate at T=(288.15 to 318.15) K: A volumetric and viscometric approach. | Banipal, T. S., Kaur, J., & Banipal, P. K. (2012). 2012. The Journal of Chemical Thermodynamics. 48: 181-189.
- Synthesis of saucer shaped manganese oxide nanoparticles by co-precipitation method and the application as fuel additive | Jamil, S., Khan, S. R., Sultana, B., Hashmi, M., Haroon, M., & Janjua, M. R. S. A. 2018. Journal of Cluster Science. 29: 1099-1106.
- Removal of Zn (II) from manganese-zinc chloride waste liquor using ion-exchange with D201 resin. | Zhou, K., Wu, Y., Zhang, X., Peng, C., Cheng, Y., & Chen, W. 2019. Hydrometallurgy. 190: 105171.
- Tailoring manganese oxide nanoplates enhances oxygen evolution catalysis in acid. | Zhao, Z., Zhang, B., Fan, D., Wang, Y., Yang, H., Huang, K.,.. & Lei, M. 2022. Journal of Catalysis. 405: 265-272.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Manganese Chloride Tetrahydrate, 100 g | sc-207841 | 100 g | $46.00 | |||
Manganese Chloride Tetrahydrate, 500 g | sc-207841A | 500 g | $77.00 |
