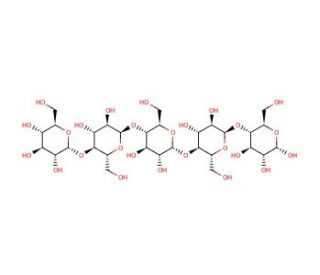
Molecular structure of Maltopentaose, CAS Number: 34620-76-3
Maltopentaose (CAS 34620-76-3)
See product citations (1)
Alternate Names:
Maltopentaose is also known as Amylopentaose.
Application:
Maltopentaose is a malto-oligosaccharide derived from starch by hydrolysis and chromatography for oligosaccharide research and diagnostics.
CAS Number:
34620-76-3
Purity:
≥90%
Molecular Weight:
828.72
Molecular Formula:
C30H52O26
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Product Citations
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Maltopentaose is an oligosaccharide for oligosaccharide research and diagnostics. Maltopentaose is a malto-oligosaccharide comprised of five α-1,4-linked glucose molecules. Maltopentaose increases the α-amylase synthesis rate in B. stearothermophilus when used at a concentration of 1 mM. Maltopentaose has been used as a substrate for porcine pancreatic α-amylase (PPA) to study various inhibitors of PPA. Maltopentaose is also known as Maltopentose, O-α-D-glucopyranosyl-(1→4)-O-α-D-glucopyranosyl-(1→4)-O-α-D-glucopyranosyl-(1→4)-O-α-D-glucopyranosyl-(1→4)-D-glucose, and Amylopentaose.
Maltopentaose (CAS 34620-76-3) References
- Mechanism of porcine pancreatic alpha-amylase. Inhibition of amylose and maltopentaose hydrolysis by alpha-, beta- and gamma-cyclodextrins. | Koukiekolo, R., et al. 2001. Eur J Biochem. 268: 841-8. PMID: 11168426
- Crystal structure of a catalytic site mutant of beta-amylase from Bacillus cereus var. mycoides cocrystallized with maltopentaose. | Miyake, H., et al. 2003. Biochemistry. 42: 5574-81. PMID: 12741813
- Cellodextrin preparation by mixed-acid hydrolysis and chromatographic separation. | Zhang, YH. and Lynd, LR. 2003. Anal Biochem. 322: 225-32. PMID: 14596831
- Site-directed mutagenesis of the greasy slide aromatic residues within the LamB (maltoporin) channel of Escherichia coli: effect on ion and maltopentaose transport. | Denker, K., et al. 2005. J Mol Biol. 352: 534-50. PMID: 16095613
- Crystal structure of the rice branching enzyme I (BEI) in complex with maltopentaose. | Chaen, K., et al. 2012. Biochem Biophys Res Commun. 424: 508-11. PMID: 22771800
- A novel electrochemical method to determine α-amylase activity. | Zhang, J., et al. 2014. Analyst. 139: 3429-33. PMID: 24855635
- Importance of Trp139 in the product specificity of a maltooligosaccharide-forming amylase from Bacillus stearothermophilus STB04. | Xie, X., et al. 2019. Appl Microbiol Biotechnol. 103: 9433-9442. PMID: 31676918
- Carbohydrate-Binding Module and Linker Allow Cold Adaptation and Salt Tolerance of Maltopentaose-Forming Amylase From Marine Bacterium Saccharophagus degradans 2-40 T. | Ding, N., et al. 2021. Front Microbiol. 12: 708480. PMID: 34335544
- Fusion of maltooligosaccharide-forming amylases from two origins for the improvement of maltopentaose synthesis. | Han, X., et al. 2021. Food Res Int. 150: 110735. PMID: 34865754
- Liquid chromatographic assay of the relative activities of serum pancreatic and salivary alpha-amylase using reductively pyridylaminated maltopentaose as a fluorescent substrate. | Honda, S., et al. 1987. J Chromatogr. 419: 51-60. PMID: 3499444
- Starch-Binding Domain Modulates the Specificity of Maltopentaose Production at Moderate Temperatures. | Ding, N., et al. 2022. J Agric Food Chem. 70: 9057-9065. PMID: 35829707
- Sensitive detection and structural characterization of trimethyl(p-aminophenyl)-ammonium-derivatized oligosaccharides by electrospray ionization-mass spectrometry and tandem mass spectrometry. | Okamoto, M., et al. 1995. Rapid Commun Mass Spectrom. 9: 641-3. PMID: 7647361
- Crystal structures of a mutant maltotetraose-forming exo-amylase cocrystallized with maltopentaose. | Yoshioka, Y., et al. 1997. J Mol Biol. 271: 619-28. PMID: 9281429
- Effect of pressure on the mechanism of hydrolysis of maltotetraose, maltopentaose, and maltohexose catalyzed by porcine pancreatic alpha-amylase. | Matsumoto, T., et al. 1997. Biochim Biophys Acta. 1343: 243-50. PMID: 9434115
- Crystal structure of a catalytic-site mutant alpha-amylase from Bacillus subtilis complexed with maltopentaose. | Fujimoto, Z., et al. 1998. J Mol Biol. 277: 393-407. PMID: 9514750
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Maltopentaose, 25 mg | sc-218666C | 25 mg | $180.00 | |||
Maltopentaose, 50 mg | sc-218666 | 50 mg | $240.00 | |||
Maltopentaose, 100 mg | sc-218666A | 100 mg | $480.00 | |||
Maltopentaose, 250 mg | sc-218666D | 250 mg | $800.00 | |||
Maltopentaose, 500 mg | sc-218666E | 500 mg | $1400.00 | |||
Maltopentaose, 1 g | sc-218666B | 1 g | $2600.00 |
