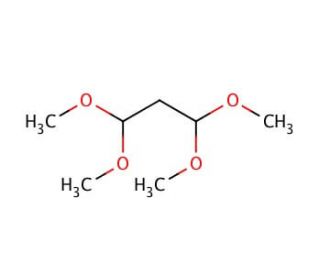
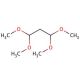
Malonaldehyde bis-(dimethyl acetal) (CAS 102-52-3)
QUICK LINKS
Malonaldehyde bis-(dimethyl acetal) is a chemical compound that functions as a cross-linking agent in various experimental applications. Its mechanism of action involves reacting with functional groups on biomolecules, such as proteins and nucleic acids, to form stable cross-links. This process can be utilized to study the structure and function of biomolecules, as well as to investigate protein-protein or protein-nucleic acid interactions. Malonaldehyde bis-(dimethyl acetal) can be used to stabilize and preserve biological samples for further analysis, as it can effectively cross-link and immobilize biomolecules. Its ability to form stable cross-links may be a useful for studying the molecular interactions and stability of biological samples in research and development applications.
Malonaldehyde bis-(dimethyl acetal) (CAS 102-52-3) References
- Lipid peroxidation-DNA damage by malondialdehyde. | Marnett, LJ. 1999. Mutat Res. 424: 83-95. PMID: 10064852
- Kinetics of tumor necrosis factor alpha in plasma and the cardioprotective effect of a monoclonal antibody to tumor necrosis factor alpha in acute myocardial infarction. | Li, D., et al. 1999. Am Heart J. 137: 1145-52. PMID: 10347344
- Chemistry and biology of DNA damage by malondialdehyde. | Marnett, LJ. 1999. IARC Sci Publ. 17-27. PMID: 10626205
- Comparison of IY81149 with omeprazole in rat reflux oesophagitis. | Kil, BJ., et al. 2000. J Auton Pharmacol. 20: 291-6. PMID: 11350494
- The effect of luteolin-7-O-beta-D-glucuronopyranoside on gastritis and esophagitis in rats. | Min, YS., et al. 2006. Arch Pharm Res. 29: 484-9. PMID: 16833016
- Synergism between aflatoxins in covalent binding to DNA and in mutagenesis in the photoactivation system. | Stark, AA. and Liberman, DF. 1991. Mutat Res. 247: 77-86. PMID: 1900569
- Melanosis and quality changes of Pacific white shrimp (Litopenaeus vannamei) treated with catechin during iced storage. | Nirmal, NP. and Benjakul, S. 2009. J Agric Food Chem. 57: 3578-86. PMID: 19309075
- Chemistry and biochemistry of 4-hydroxynonenal, malonaldehyde and related aldehydes. | Esterbauer, H., et al. 1991. Free Radic Biol Med. 11: 81-128. PMID: 1937131
- Hydrogen peroxide modifies human sperm peroxiredoxins in a dose-dependent manner. | O'Flaherty, C. and de Souza, AR. 2011. Biol Reprod. 84: 238-47. PMID: 20864641
- Oxidized LDL attenuates protective autophagy and induces apoptotic cell death of endothelial cells: Role of oxidative stress and LOX-1 receptor expression. | Mollace, V., et al. 2015. Int J Cardiol. 184: 152-158. PMID: 25703423
- In vivo oxidative stress alters thiol redox status of peroxiredoxin 1 and 6 and impairs rat sperm quality. | Liu, Y. and O'Flaherty, C. 2017. Asian J Androl. 19: 73-79. PMID: 26823067
- High-performance liquid chromatography analysis of the thiobarbituric acid adducts of malonaldehyde and trans,trans-muconaldehyde. | Yu, LW., et al. 1986. Anal Biochem. 156: 326-33. PMID: 3766936
- Glutathione-dependent protection by rat liver microsomal protein against lipid peroxidation. | Burk, RF. 1983. Biochim Biophys Acta. 757: 21-8. PMID: 6838905
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Malonaldehyde bis-(dimethyl acetal), 5 ml | sc-202219 | 5 ml | $47.00 |
