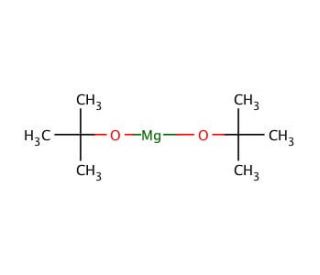
Molecular structure of Magnesium di-tert-butoxide, CAS Number: 32149-57-8
Magnesium di-tert-butoxide (CAS 32149-57-8)
Alternate Names:
Magnesium tert-butoxide
CAS Number:
32149-57-8
Molecular Weight:
170.53
Molecular Formula:
(CH3)3COMgOC(CH3)3
Supplemental Information:
This is classified as a Dangerous Good for transport and may be subject to additional shipping charges.
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Magnesium di-tert-butoxide is used as a reagent for deprotonation processes within organic synthesis. It is a mild alkali-earth metal alkoxide base, simultaneously operating as a Lewis acid and a Bronsted base in chemical transformations. It is used in the synthesis of acyclic nucleoside phosphonates and as a catalyst for diverse organic reactions, such as hydrolysis. This organometallic compound, Magnesium di-tert-butoxide, reacts with alcohols, thiols, amines, and carboxylic acids, yielding Grignard reagents.
Magnesium di-tert-butoxide (CAS 32149-57-8) References
- Cooperative catalysis by carbenes and Lewis acids in a highly stereoselective route to gamma-lactams. | Raup, DE., et al. 2010. Nat Chem. 2: 766-71. PMID: 20729898
- Ester prodrugs of acyclic nucleoside thiophosphonates compared to phosphonates: synthesis, antiviral activity and decomposition study. | Roux, L., et al. 2013. Eur J Med Chem. 63: 869-81. PMID: 23603046
- A cooperative N-heterocyclic carbene/chiral phosphate catalysis system for allenolate annulations. | Lee, A. and Scheidt, KA. 2014. Angew Chem Int Ed Engl. 53: 7594-8. PMID: 24895280
- Synthesis and chemical reactivity of a 6-Me-3,2-hydroxypyridinone dithiazolide with primary amines: a route to new hexadentate chelators for hard metal(III) ions. | Pailloux, SL., et al. 2016. J Heterocycl Chem. 53: 1065-1073. PMID: 27667855
- A Cooperative Ternary Catalysis System for Asymmetric Lactonizations of α-Ketoesters. | Murauski, KJR., et al. 2017. Adv Synth Catal. 359: 3713-3719. PMID: 30473657
- Therapeutic use of fluorinated nucleosides - progress in patents. | Bassetto, M. and Slusarczyk, M. 2018. Pharm Pat Anal. 7: 277-299. PMID: 30657414
- New Developments of the Principle of Vinylogy as Applied to π-Extended Enolate-Type Donor Systems. | Curti, C., et al. 2020. Chem Rev. 120: 2448-2612. PMID: 32040305
- 1,2,4-Thiadiazole acyclic nucleoside phosphonates as inhibitors of cysteine dependent enzymes cathepsin K and GSK-3β. | Pomeislová, A., et al. 2021. Bioorg Med Chem. 32: 115998. PMID: 33440320
- New synthetic routes to nano-composites with ceramic particles, using lanthanide compounds | Veith, M. 2008. Journal of sol-gel science and technology. 46: 291-298.
- An Efficient Synthesis of N‐(Hetero) arylcarbazoles: Palladium‐Catalyzed Coupling Reaction between (Hetero) aryl Chlorides and N‐Carbazolylmagnesium Chloride | Nakayama, Y., Yokoyama, N., Nara, H., Kobayashi, T., & Fujiwhara, M. 2015. Advanced Synthesis & Catalysis. 357(10): 2322-2330.
- Force‐Reversible and Energetic Indole‐Mg‐Indole Cation‐π Interaction for Designing Toughened and Multifunctional High‐Performance Thermosets | Yang, L., Li, Y., Du, M., He, Y., Lan, Y., Yin, Q.,.. & Chang, G. 2022. Advanced Functional Materials. 32(14): 2111021.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Magnesium di-tert-butoxide, 50 g | sc-250277 | 50 g | $192.00 |
