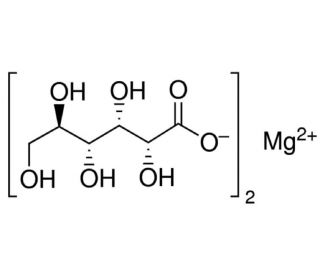
Magnesium D-gluconate (CAS 3632-91-5)
QUICK LINKS
Magnesium D-gluconate is a compound formed by the combination of magnesium ions with D-gluconate, a derivative of gluconic acid, known for its high solubility and bioavailability. This characteristic makes it a valuable tool in biochemical research, particularly in studies investigating the essential roles of magnesium in biological systems. Magnesium is critical for numerous cellular processes, including acting as a cofactor for various enzymes, facilitating DNA and RNA synthesis, and playing a pivotal role in energy metabolism through its involvement in ATP synthesis. The stability and solubility of Magnesium D-gluconate allow precise control and manipulation of intracellular magnesium levels in experimental settings, enabling researchers to explore how magnesium affects enzyme activity, ion transport, cellular signaling, and neuromuscular functions. Through such studies, Magnesium D-gluconate helps explain the fundamental mechanisms by which magnesium contributes to cellular homeostasis and metabolic regulation. This compound thus serves as a key agent in scientific investigations that seek to deepen our understanding of mineral homeostasis in biological systems without the direct aim of therapeutic application.
Magnesium D-gluconate (CAS 3632-91-5) References
- Physiological basis of the low calcium response in Yersinia pestis. | Fowler, JM. and Brubaker, RR. 1994. Infect Immun. 62: 5234-41. PMID: 7960099
- Solubilities of magnesium-l-ascorbate, calcium-l-ascorbate, magnesium-l-glutamate, magnesium-d-gluconate, calcium-d-gluconate, calcium-d-heptagluconate, l-aspartic acid, and 3-nitrobenzoic acid in water | Mishelevich, A., & Apelblat, A. 2008. The Journal of Chemical Thermodynamics. 40(5): 897-900.
- Understanding the effect of inert support on the reactivity stabilization for synthetic calcium based sorbents | Li, Z., Liu, Y., & Cai, N. 2013. Chemical engineering science. 89: 235-243.
- Effects of various magnesium salts for the production of milk fermented by Bifidobacterium animalis ssp. lactis Bb-12 | Szajnar, K., Znamirowska, A., & Kalicka, D. 2019. International journal of food properties. 22(1): 1087-1099.
- High-performance CaO-based composites synthesized using a space-confined chemical vapor deposition strategy for thermochemical energy storage | Han, R., Gao, J., Wei, S., Su, Y., Su, C., Li, J.,.. & Qin, Y. 2020. Solar Energy Materials and Solar Cells. 206: 110346.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Magnesium D-gluconate, 100 g | sc-221868 | 100 g | $53.00 | |||
Magnesium D-gluconate, 500 g | sc-221868A | 500 g | $216.00 |
