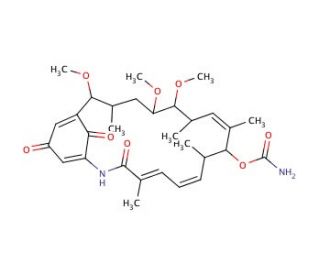
Macbecin I (CAS 73341-72-7)
See product citations (1)
QUICK LINKS
Macbecin I, registered under CAS number 73341-72-7, is a macrocyclic compound that belongs to the ansamycin class of antibiotics, which are known for their complex structures involving a cyclic lactam ring linked to an aromatic moiety. This compound is naturally produced by the bacterium Micromonospora species. Macbecin I functions primarily by inhibiting bacterial protein synthesis, specifically targeting the bacterial RNA polymerase, a crucial enzyme responsible for transcribing DNA into RNA. The unique mechanism of action involves binding to the RNA polymerase, thereby obstructing the RNA synthesis pathway and ultimately preventing bacterial growth and multiplication. In research environments, macbecin I has been utilized as a tool to study bacterial transcription mechanisms and the effects of transcription inhibition on bacterial physiology. Its ability to selectively bind and inhibit RNA polymerase has provided insights into the structural and functional aspects of this essential enzyme, which is pivotal for understanding bacterial gene expression and regulation. Furthermore, studies involving macbecin I contribute to the development of novel approaches for targeting bacterial transcription in the quest to address antibiotic resistance, offering a foundational understanding that could guide future antimicrobial strategies.
Macbecin I (CAS 73341-72-7) References
- Hsp90 binds and regulates Gcn2, the ligand-inducible kinase of the alpha subunit of eukaryotic translation initiation factor 2 [corrected]. | Donzé, O. and Picard, D. 1999. Mol Cell Biol. 19: 8422-32. PMID: 10567567
- Strategies and tactics for the synthesis of oxygenated natural products. | Martin, SF. 1992. J Nat Prod. 55: 1718-31. PMID: 1294694
- Studies on the syntheses of benzoquinone ansamycin antibiotics. Syntheses of the C5-C15 subunits of macbecin I, geldanamycin, and herbimycin A. | Belardi, JK. and Micalizio, GC. 2006. Org Lett. 8: 2409-12. PMID: 16706538
- Total synthesis of macbecin I. | Belardi, JK. and Micalizio, GC. 2008. Angew Chem Int Ed Engl. 47: 4005-8. PMID: 18404757
- Reaction design, discovery, and development as a foundation to function-oriented synthesis. | Micalizio, GC. and Hale, SB. 2015. Acc Chem Res. 48: 663-73. PMID: 25668752
- Antitumor and cytocidal activities of a newly isolated benzenoid ansamycin, macbecin I. | Ono, Y., et al. 1982. Gan. 73: 938-44. PMID: 6186564
- Macbecins I and II, new antitumor antibiotics. I. Producing organism, fermentation and antimicrobial activities. | Tanida, S., et al. 1980. J Antibiot (Tokyo). 33: 199-204. PMID: 7380729
- Macbecins I and II, new antitumor antibiotics. II. Isolation and characterization. | Muroi, M., et al. 1980. J Antibiot (Tokyo). 33: 205-12. PMID: 7380730
- Mutant conformation of p53 translated in vitro or in vivo requires functional HSP90. | Blagosklonny, MV., et al. 1996. Proc Natl Acad Sci U S A. 93: 8379-83. PMID: 8710879
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Macbecin I, 1 mg | sc-204067 | 1 mg | $245.00 |
