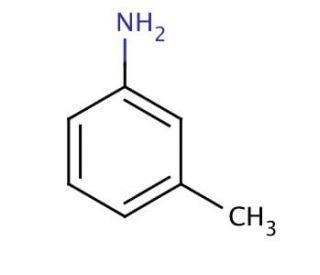
Molecular structure of m-Toluidine, CAS Number: 108-44-1
m-Toluidine (CAS 108-44-1)
Alternate Names:
3-Methylaniline
CAS Number:
108-44-1
Purity:
≥98%
Molecular Weight:
107.15
Molecular Formula:
C7H9N
Supplemental Information:
This is classified as a Dangerous Good for transport and may be subject to additional shipping charges.
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
m-Toluidine is a chemical compound that functions as a reagent in various chemical reactions. It acts as a precursor in the synthesis of dyes and other organic compounds. Its mechanism of action involves participating in nucleophilic aromatic substitution reactions, where it undergoes substitution with other compounds to form new chemical products. m-Toluidine plays a role in the modification of organic molecules. At the molecular level, m-Toluidine interacts with other reagents to facilitate the formation of complex organic structures.
m-Toluidine (CAS 108-44-1) References
- Biological monitoring of phenmedipham: determination of m-toluidine in urine. | Schettgen, T., et al. 2001. Arch Toxicol. 75: 145-9. PMID: 11409536
- Structures of mutagens produced by the co-mutagen norharman with o- and m-toluidine isomers. | Hada, N., et al. 2001. Mutat Res. 493: 115-26. PMID: 11516721
- Dynamics of glassy and liquid m-toluidine investigated by high-resolution dielectric spectroscopy. | Mandanici, A., et al. 2005. J Chem Phys. 122: 84508. PMID: 15836064
- Slow dynamics of supercooled m-toluidine investigated by mechanical spectroscopy. | Mandanici, A., et al. 2005. J Chem Phys. 122: 114501. PMID: 15836223
- Effect of dietary fat on selected parameters of toxicity following 1- or 3-month exposure of rats to toluidine isomers. | Jodynis-Liebert, J. and Bennasir, HA. 2005. Int J Toxicol. 24: 365-76. PMID: 16257856
- Density and confinement effects of glass forming m-toluidine in nanoporous Vycor investigated by depolarized dynamic light scattering. | Blochowicz, T., et al. 2013. J Chem Phys. 138: 114501. PMID: 23534643
- Integrated adsorptive technique for efficient recovery of m-cresol and m-toluidine from actual acidic and salty wastewater. | Chen, D., et al. 2016. J Hazard Mater. 312: 192-199. PMID: 27037473
- Biodegradation of phenmedipham by novel Ochrobactrum anthropi NC-1. | Pujar, NK., et al. 2019. 3 Biotech. 9: 52. PMID: 30729076
- Improvement of the adhesion of conductive poly(m-toluidine) onto chemically reduced-wool fabrics. | Kalkan ErdoĞan, M., et al. 2020. Turk J Chem. 44: 775-790. PMID: 33488193
- Biochar-mediated reduction of m-nitrotoluene: Interaction between reduction of m-nitrotoluene and sequestration of contaminants. | Wu, L., et al. 2021. Sci Total Environ. 773: 145662. PMID: 33940750
- Fabrication, Modification, and Characterization of Lignin-Based Electrospun Fibers Derived from Distinctive Biomass Sources. | Attia, AAM., et al. 2021. Polymers (Basel). 13: PMID: 34301035
- Effect of Au Plasmonic Material on Poly M-Toluidine for Photoelectrochemical Hydrogen Generation from Sewage Water. | Abdelazeez, AAA., et al. 2022. Polymers (Basel). 14: PMID: 35215683
- Discolouring 3D Gel Dosimeter for UV Dose Distribution Measurements. | Jaszczak, M., et al. 2022. Materials (Basel). 15: PMID: 35407878
- Exposure to melamine and its derivatives and aromatic amines among pregnant women in the United States: The ECHO Program. | Choi, G., et al. 2022. Chemosphere. 307: 135599. PMID: 36055588
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
m-Toluidine, 5 ml | sc-250270 | 5 ml | $30.00 | |||
m-Toluidine, 100 ml | sc-250270A | 100 ml | $50.00 |
