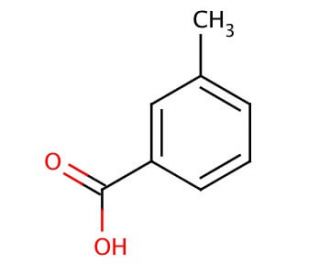
m-Toluic acid (CAS 99-04-7)
QUICK LINKS
m-Toluic acid is a chemical compound that functions as a precursor in the synthesis of various organic compounds. It acts as a building block for the production of dyes and perfumes. Its mechanism of action involves participating in organic reactions, such as esterification, amidation, and oxidation, to form more complex molecules. In these reactions, m-Toluic acid serves as a starting material, providing the necessary functional groups for the formation of new chemical entities. Its role in the experimental context is to facilitate the creation of diverse organic compounds through its reactivity and ability to undergo various chemical transformations. At the molecular level, m-Toluic acid interacts with other reagents and catalysts to enable the formation of desired products, contributing to the advancement of chemistry research.
m-Toluic acid (CAS 99-04-7) References
- Simultaneous removal of phenol, ortho- and para-cresol by mixed anaerobic consortia. | Tawfiki Hajji, K., et al. 1999. Can J Microbiol. 45: 318-25. PMID: 10490333
- Effects of the headspace gas composition on anaerobic biotransformation of o-, m-, and p-toluic acid in sediment slurries. | Liu, SM. and Chi, WC. 2003. J Environ Sci Health A Tox Hazard Subst Environ Eng. 38: 1099-113. PMID: 12774912
- MICROBIAL METABOLISM OF AROMATIC COMPOUNDS. I. DECOMPOSITION OF PHENOLIC COMPOUNDS AND AROMATIC HYDROCARBONS BY PHENOL-ADAPTED BACTERIA. | TABAK, HH., et al. 1964. J Bacteriol. 87: 910-9. PMID: 14137630
- N,N-diethyl phenylacetamide (DEPA): A safe and effective repellent for personal protection against hematophagous arthropods. | Kalyanasundaram, M. and Mathew, N. 2006. J Med Entomol. 43: 518-25. PMID: 16739410
- Gene targeting in gram-negative bacteria by use of a mobile group II intron ('Targetron') expressed from a broad-host-range vector. | Yao, J. and Lambowitz, AM. 2007. Appl Environ Microbiol. 73: 2735-43. PMID: 17322321
- Chromatographic fingerprints of industrial toluic acids established for their quality control. | Lian, H. and Wei, Y. 2007. Talanta. 71: 264-9. PMID: 19071298
- Fe protein over-expression can enhance the nitrogenase activity of Azotobacter vinelandii. | Nag, P. and Pal, S. 2013. J Basic Microbiol. 53: 156-62. PMID: 22581696
- Benzimidazole or Diamide From a Reaction of Diamines and Carboxylic Acids or Acid Chlorides: Crystal Structures and Theoretical Studies. | Odame, F., et al. 2015. Acta Chim Slov. 62: 986-94. PMID: 26680729
- Comparative Single-Cell Analysis of Different E. coli Expression Systems during Microfluidic Cultivation. | Binder, D., et al. 2016. PLoS One. 11: e0160711. PMID: 27525986
- A novel expression system for lytic polysaccharide monooxygenases. | Courtade, G., et al. 2017. Carbohydr Res. 448: 212-219. PMID: 28291518
- Switching the Ligand Specificity of the Biosensor XylS from meta to para-Toluic Acid through Directed Evolution Exploiting a Dual Selection System. | Ogawa, Y., et al. 2019. ACS Synth Biol. 8: 2679-2689. PMID: 31689072
- Development of Nickel-Based Negative Tone Metal Oxide Cluster Resists for Sub-10 nm Electron Beam and Helium Ion Beam Lithography. | Kumar, R., et al. 2020. ACS Appl Mater Interfaces. 12: 19616-19624. PMID: 32267144
- Engineering of the Ligand Specificity of Transcriptional Regulator XylS by Deep Mutational Scanning. | Ogawa, Y., et al. 2022. ACS Synth Biol. 11: 473-485. PMID: 34964613
- Controllable fluorescence via tuning the m-substituents of added aromatic molecules in a pyrene derivative-decorated porous skeleton. | Wu, J., et al. 2019. RSC Adv. 9: 20185-20191. PMID: 35514678
- Thermochemical investigations of nearly ideal binary solvents. VII: Monomer and dimer models for solubility of benzoic acid in simple binary and ternary solvents. | Acree, WE. and Bertrand, GL. 1981. J Pharm Sci. 70: 1033-6. PMID: 6101149
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
m-Toluic acid, 5 g | sc-250269 | 5 g | $20.00 | |||
m-Toluic acid, 100 g | sc-250269A | 100 g | $42.00 |
