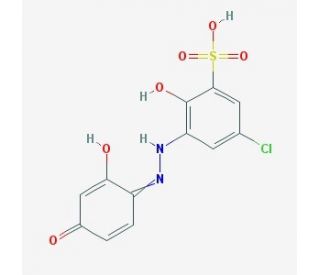
Lumogallion (CAS 4386-25-8)
See product citations (5)
QUICK LINKS
Lumogallion is a chemical compound with a distinctive fluorescent property. It is often used as a chelating agent and colorimetric indicator for metal ions, particularly aluminum. Lumogallion has a strong affinity for aluminum ions, forming a stable complex with a characteristic red color. In terms of its chemical structure, lumogallion consists of a naphthalene ring fused with a benzene ring, and an azine group (-N=N-) attached to the naphthalene ring. This arrangement gives lumogallion its unique properties, including its ability to selectively bind with certain metal ions. One of the significant applications of lumogallion is in the detection and quantification of aluminum ions in various samples. Due to its high selectivity for aluminum, lumogallion can be used as a visual indicator to determine the presence and concentration of aluminum ions. The complex formed between lumogallion and aluminum exhibits a distinct red color, allowing for easy identification and measurement. Lumogallion has found utility in various fields, including environmental analysis, biomedical research, and industrial processes. It has been employed in studies investigating aluminum toxicity, aluminum contamination in water sources, and the assessment of aluminum content in biological samples. In addition to its aluminum-specific properties, lumogallion has also been investigated for its potential use as a fluorescent probe for other metal ions, such as zinc and copper. These applications rely on the ability of lumogallion to form complexes with these metals and exhibit fluorescence emission at specific wavelengths. It is worth noting that lumogallion may have limitations, including its selectivity for aluminum and potential interference from other metal ions. Therefore, proper controls and calibration are necessary when using lumogallion for quantitative analysis. In summary, lumogallion is a chemical compound employed as a chelating agent and colorimetric indicator for aluminum ions. Its distinctive red color upon complex formation with aluminum makes it useful in various analytical applications. Ongoing research aims to further explore its potential uses and optimize its performance in detecting and quantifying metal ions.
Lumogallion (CAS 4386-25-8) References
- Determination of aluminum in beverages by automated non-segmented continuous flow analysis with fluorescent detection of the lumogallion complex. | Hoch, RL. 1999. Analyst. 124: 793-6. PMID: 10616741
- Aluminum accumulation at nuclei of cells in the root tip. Fluorescence detection using lumogallion and confocal laser scanning microscopy. | Silva, IR., et al. 2000. Plant Physiol. 123: 543-52. PMID: 10859184
- Speciation of aluminum in soil extracts using cation and anion exchangers followed by a flow-injection system with fluorescence detection using lumogallion. | Yamada, E., et al. 2002. Anal Sci. 18: 785-91. PMID: 12137374
- High performance liquid chromatographic determination of aluminium in natural waters in the form of its lumogallion chelate. | Zhou, CY., et al. 1995. Talanta. 42: 415-22. PMID: 18966246
- Fully-automated fluorimetric determination of aluminum in seawater by in-syringe dispersive liquid-liquid microextraction using lumogallion. | Suárez, R., et al. 2012. Anal Chem. 84: 9462-9. PMID: 23005696
- Aluminum content of human semen: implications for semen quality. | Klein, JP., et al. 2014. Reprod Toxicol. 50: 43-8. PMID: 25461904
- Al adjuvants can be tracked in viable cells by lumogallion staining. | Mile, I., et al. 2015. J Immunol Methods. 422: 87-94. PMID: 25896212
- Insight into the cellular fate and toxicity of aluminium adjuvants used in clinically approved human vaccinations. | Mold, M., et al. 2016. Sci Rep. 6: 31578. PMID: 27515230
- Boron increases root elongation by reducing aluminum induced disorganized distribution of HG epitopes and alterations in subcellular cell wall structure of trifoliate orange roots. | Riaz, M., et al. 2018. Ecotoxicol Environ Saf. 165: 202-210. PMID: 30196002
- Self-plasticized, lumogallion-based fluorescent optical sensor for the determination of aluminium (III) with ultra-low detection limits. | Mendecki, L., et al. 2020. Anal Chim Acta. 1101: 141-148. PMID: 32029105
- Determination of serum and urinary aluminum by HPLC with fluorometric detection of Al-lumogallion complex. | Lee, BL., et al. 1996. Clin Chem. 42: 1405-11. PMID: 8787696
- Sensitive detection of trace aluminium in biological tissues by confocal laser scanning microscopy after staining with lumogallion. | Uchiumi, A., et al. 1998. Analyst. 123: 759-62. PMID: 9684408
- Staining for histologic diagnosis of aluminum osteopathy--application of confocal laser scanning microscopy to observation for offminum in bones from aluminum-treated, subtotally nephrectomized rats. | Teraki, Y., et al. 1998. Kaibogaku Zasshi. 73: 317-28. PMID: 9778918
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Lumogallion, 100 mg | sc-295368 | 100 mg | $107.00 | |||
Lumogallion, 1 g | sc-295368A | 1 g | $169.00 |
