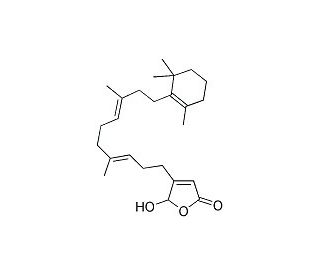
Molecular structure of Luffariellolide, CAS Number: 111149-87-2
Luffariellolide (CAS 111149-87-2)
Application:
Luffariellolide is an anti-inflammatory PLA2 inhibitor
CAS Number:
111149-87-2
Purity:
≥98%
Molecular Weight:
386.57
Molecular Formula:
C25H38O3
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Luffariellolide is structurally related to manoalide. Luffariellolide is an anti-inflammatory PLA2 (phospholipase A2) inhibitor (IC50= 0.23 muM). This compound is less potent than manoalide, but partially reversible. Luffariellolide inhibits human recombinant sPLA2 selectively over cPLA2.
Luffariellolide (CAS 111149-87-2) References
- Dysidiolide and related gamma-hydroxy butenolide compounds as inhibitors of the protein tyrosine phosphatase, CDC25. | Blanchard, JL., et al. 1999. Bioorg Med Chem Lett. 9: 2537-8. PMID: 10498203
- Phospholipase A2 inhibition by alkylbenzoylacrylic acids. | Köhler, T., et al. 1992. Biochem Pharmacol. 44: 805-13. PMID: 1324685
- Characterization and pharmacological modulation of soluble phospholipase A2 generated during glycogen-induced rat peritonitis. | Marshall, LA., et al. 1992. Agents Actions. 37: 60-9. PMID: 1456181
- Are prostanoids related to positive inotropism by UTP and ATP? | Froldi, G., et al. 2005. Pharmacology. 73: 140-5. PMID: 15564788
- New luffariellolide derivatives from the Indonesian sponge Acanthodendrilla sp. | Elkhayat, E., et al. 2004. J Nat Prod. 67: 1809-17. PMID: 15568767
- PAF and LTB4 biosynthesis in the human neutrophil: effects of putative inhibitors of phospholipase A2 and specific inhibitors of 5-lipoxygenase. | Glaser, KB., et al. 1991. Agents Actions. 34: 89-92. PMID: 1665306
- Bioactive sesterterpenes and triterpenes from marine sponges: occurrence and pharmacological significance. | Ebada, SS., et al. 2010. Mar Drugs. 8: 313-46. PMID: 20390108
- Revealing a natural marine product as a novel agonist for retinoic acid receptors with a unique binding mode and inhibitory effects on cancer cells. | Wang, S., et al. 2012. Biochem J. 446: 79-87. PMID: 22642567
- Structures and potential antitumor activity of sesterterpenes from the marine sponge Hyrtios communis. | Li, J., et al. 2013. J Nat Prod. 76: 1492-7. PMID: 23944963
- Sesterterpenoids with Anticancer Activity. | Evidente, A., et al. 2015. Curr Med Chem. 22: 3502-22. PMID: 26295461
- Pharmacologic modulation of D-49 phospholipase A2-induced paw edema in the mouse. | Calhoun, W., et al. 1989. Agents Actions. 27: 418-21. PMID: 2801333
- Dactylospenes A-E, Sesterterpenes from the Marine Sponge Dactylospongia elegans. | Yu, HB., et al. 2020. Mar Drugs. 18: PMID: 32993037
- 5-hydroxy-3-vinyl-2(5H)-furanone--a new inhibitor of human synovial phospholipase A2 and platelet aggregation from fermentations of a Calyptella species (basidiomycetes). | Lorenzen, K., et al. 1995. Z Naturforsch C J Biosci. 50: 403-9. PMID: 7546033
- Cellular and topical in vivo inflammatory murine models in the evaluation of inhibitors of phospholipase A2. | Glaser, KB., et al. 1995. Skin Pharmacol. 8: 300-8. PMID: 8688196
Inhibitor of:
Phospholipase Activating peptides and, PLA2, and sPLA2.Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Luffariellolide, 1 mg | sc-202212 | 1 mg | $263.00 |
