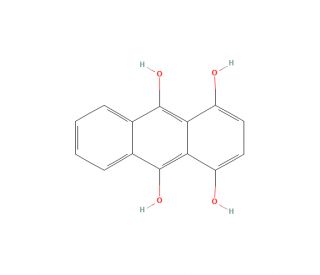
Leucoquinizarin (CAS 476-60-8)
QUICK LINKS
Leucoquinizarin is a naturally occurring quinone compound and a precursor to several anthraquinone derivatives. It is found in many plant species, including the species of the genus Rubus, and is widely used in the fields of biochemistry and pharmaceutical research. Leucoquinizarin acts as an electron acceptor in biochemical reactions. It can be used to measure the rate of these reactions, as well as to study the structure and function of proteins and DNA. In addition, Leucoquinizarin may act to inhibit the growth of certain types of cancer cells and reduce the toxicity of certain drugs. Furthermore, it can increase the production of certain enzymes, such as tyrosinase and catalase, which are important for the metabolism of proteins and other molecules.
Leucoquinizarin (CAS 476-60-8) References
- The structure of anthracycline derivatives determines their subcellular localization and cytotoxic activity. | Shaul, P., et al. 2013. ACS Med Chem Lett. 4: 323-8. PMID: 24900668
- [Total synthesis of oxa-9-anthracyclines]. | Dufat-Trinh Van, H., et al. 1989. Chem Pharm Bull (Tokyo). 37: 3294-300. PMID: 2632074
- Energy of Intramolecular Hydrogen Bonding in ortho-Hydroxybenzaldehydes, Phenones and Quinones. Transfer of Aromaticity from ipso-Benzene Ring to the Enol System(s). | Rusinska-Roszak, D. 2017. Molecules. 22: PMID: 28335484
- Keto-enol tautomerization drives the self-assembly of leucoquinizarin on Au(111). | Costantini, R., et al. 2020. Chem Commun (Camb). 56: 2833-2836. PMID: 32065182
- Enhancing the solubility of 1,4-diaminoanthraquinones in electrolytes for organic redox flow batteries through molecular modification. | Geysens, P., et al. 2020. RSC Adv. 10: 39601-39610. PMID: 35515364
- Leucoquinizarin as an analytical spectrophotometric and fluorimetric reagent: application to the determination of magnesium in pharmaceutical preparations. | Bello López, MA., et al. 1986. Analyst. 111: 429-33. PMID: 3717589
- Antineoplastic agents. Structure-activity relationship study of bis(substituted aminoalkylamino)anthraquinones. | Zee-Cheng, RK. and Cheng, CC. 1978. J Med Chem. 21: 291-4. PMID: 628005
- Studies related to anthracyclines. Part 1. Some Diels-Alder reactions of 4a,9a-epoxy-4a,9a-dihydroanthracene-1,4,9,10-tetrone. | Chandler, M. and Stoodley, RJ. 1980. J Chem Soc Perkin 1. 4: 1007-12. PMID: 7190153
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Leucoquinizarin, 25 g | sc-475556 | 25 g | $72.00 |
