Leucomycin A1 (CAS 16846-34-7)
QUICK LINKS
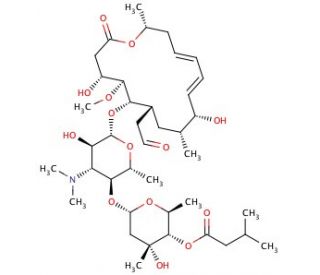
Leucomycin A1, registered under CAS number 16846-34-7, is a macrolide antibiotic that is part of a larger group of leucomycins, naturally produced by Streptomyces bacteria. This compound is characterized by a large lactone ring and multiple sugar moieties, which are critical for its mode of action and biological interactions. Leucomycin A1 functions by binding to the bacterial ribosome, specifically targeting the 50S subunit. This binding interferes with the translocation step of protein synthesis, which is crucial for the growth and replication of bacterial cells. In research contexts, Leucomycin A1 is utilized to study bacterial protein synthesis, particularly focusing on the mechanisms by which ribosomal interactions can be disrupted. It serves as a valuable tool for exploring the structural and functional dynamics of ribosomal components and their role in the synthesis of proteins. Through these studies, researchers can gain deeper insights into the process of translation inhibition and the structural requirements for antibiotic efficacy against specific bacterial targets. Additionally, Leucomycin A1 is used in studies aimed at understanding bacterial resistance mechanisms, allowing scientists to explore how bacteria evolve to overcome the inhibitory effects of macrolide antibiotics. This research is fundamental in advancing the knowledge of bacterial biology and can contribute to the development of novel strategies to mitigate resistance.
Leucomycin A1 (CAS 16846-34-7) References
- 4-O-Acetyl mycarose. A new O-acetyl sugar obtained from leucomycin minor components. | WATANABE, T., et al. 1961. J Biochem. 50: 197-201. PMID: 14005205
- Acid catalysed degradation of some spiramycin derivatives found in the antibiotic bitespiramycin. | Shi, X., et al. 2004. J Pharm Biomed Anal. 36: 593-600. PMID: 15522535
- Studies on leucomycin A1. I. Biological studies. | Hoshino, Y., et al. 1966. J Antibiot (Tokyo). 19: 23-9. PMID: 5952018
- Studies on leucomycin A1. II. Studies on blood and tissue levels, and toxicities. | Hoshino, Y., et al. 1966. J Antibiot (Tokyo). 19: 30-6. PMID: 5952019
- Studies on leucomycin A1. 3. Experimental treatments of animal infected with several pathogene bacteria. | Hoshino, Y., et al. 1966. J Antibiot (Tokyo). 19: 37-41. PMID: 5952020
- Studies on leucomycin A1. IV. Combined effects with sulfa drugs. | Hoshino, Y., et al. 1966. J Antibiot (Tokyo). 19: 42-8. PMID: 5952021
- The chemistry of the leucomycins. I. Partial structure of leucomycin A3. | Omura, S., et al. 1967. Tetrahedron Lett. 7: 609-13. PMID: 6044206
- Cloning of the macrolide antibiotic biosynthesis gene acyA, which encodes 3-O-acyltransferase, from Streptomyces thermotolerans and its use for direct fermentative production of a hybrid macrolide antibiotic. | Arisawa, A., et al. 1994. Appl Environ Microbiol. 60: 2657-60. PMID: 8074537
- Structure of leucomycin A1. | Hata and Toju, et al. 1967. Chemical and Pharmaceutical Bulletin. 15.3: 358-359.
- THE CHEMISTRY OF LEUCOMYCINS. IV STRUCTURE OF LEUCOMYCIN A 1. | Omura, et al. 1968. The Journal of Antibiotics. 21.3: 199-203.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Leucomycin A1, 1 mg | sc-391629 | 1 mg | $321.00 |
